|
 |
- Search
| J Korean Med Assoc > Volume 55(9); 2012 > Article |
Abstract
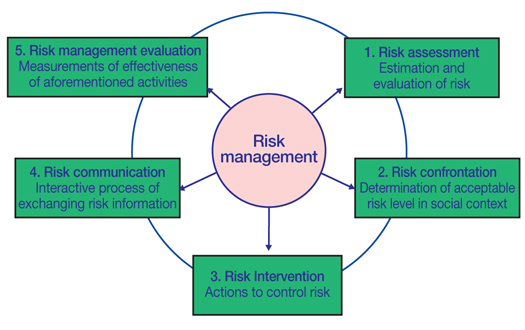
A safe drug is defined as one for which the benefit of the medication is greater than the expected risk, taking all factors into account. To enhance the safety of drugs, the risk of the drugs should be minimized. These risk management (RM) actions are assessment, confrontation, intervention, communication of risk, and evaluation of each process, in that order. To maximize the effect of RM, it should take place as early as possible in the drug life cycle, such as from the drug development period before marketing, and also after marketing. In areas of advanced drug development and a large drug market share, these activities have been legislated as Risk Evaluation and Mitigation Strategies (REMS, 2007) in the United States and the Risk Management Plan (2006) in the EU. REMS is briefly summarized in this paper. In Korea, these policies were accepted and carried out beginning in 2011 as the first stage of legal support and modification of drug safety plans. Also, proactively, the Korea Institute of Drug Safety was launched to manage information about drug safety and develop a scientific method for enhancing RM. RM should be approached in a global scope in order to foster many experts in diverse fields and to keep openness and transparency for close and effective collaboration, which is essential for successful RM and health outcomes.
References
1. Lazarou J, Pomeranz BH, Corey PN. Incidence of adverse drug reactions in hospitalized patients: a meta-analysis of prospective studies. JAMA 1998;279:1200-1205.
2. World Health Organization. WHO Collaborating Center for International Drug Monitoring. The importance of pharmacovigilance 2002;Geneva: World Health Organization.
3. Kohn LT, Corrigan J, Donaldson MS. Institute of Medicine (US) Committee on Quality of Health Care in America. To err is human: building a safer health system 2000;Washington, DC: National Academy Press.
4. Korean Society for Pharmacoepidemiology and Risk Management. Korean Society for Pharmacoepidemiology and Risk Management. Concept of risk management. Pharmacoepidemiology 2011;Seoul: Seoul National University Press. 10-15.
5. Graham DJ, Mosholder AD, Gelperin K, Avigan MI. In: Strom BL, editor. Pharmacoepidemiology and risk management. Pharmacoepidemiology 2005;4th ed. Hoboken: John Wiley & Sons. 515-530.
6. US Food and Drug Administration. Guidance for industry: development and use of risk minimization action plans [Internet] 2005;cited 2012 Aug 27. Rockville (MD): US Food and Drug Administration. Available from: http://www.fda.gov/ohrms/dockets/ac/05/briefing/2005-4136b1_03_Risk%20Minimization%20Action%20Plans.pdf
7. US Food and Drug Administration. Food and Drug Administration Amendments Act (FDAAA) of 2007 [Internet] 2007;cited 2012 Aug 27. Silver Spring (MD): US Food and Drug Administration. Available from: http://www.fda.gov/RegulatoryInformation/Legislation/FederalFoodDrugandCosmeticActFDCAct/SignificantAmendmentstotheFDCAct/FoodandDrugAdministrationAmendmentsActof2007/default.htm
8. Center for Drug Evaluation and Research (US). Center for Biologics Evaluation and Research (US). Guidance for industry: format and content of proposed risk evaluation and mitigation strategies (REMS), REMS assessments, and proposed REMS modifications 2009;Rockville (MD): US Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Center for Biologics Evaluation and Research.
Table 1
Risk management regulations
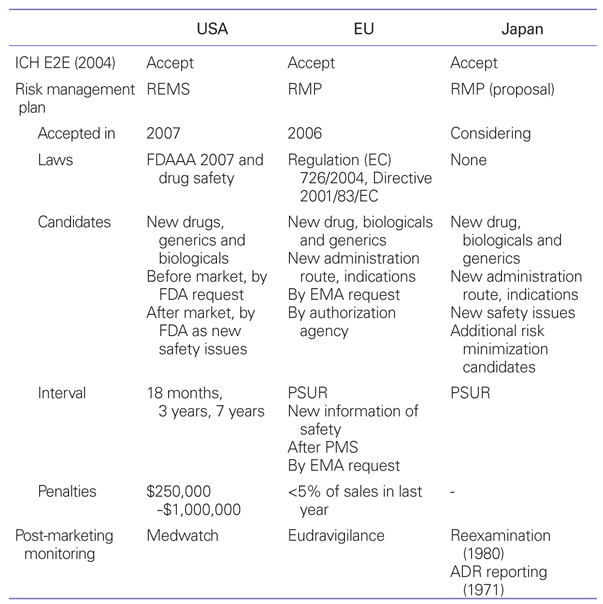
ICH E2E, International Conference on Harmonization of Technical Requirements for Registration of Pharmaceuticals for Human Use; REMS, Risk Evaluation and Mitigation Strategy; RMP, Risk Management Plans; FDAAA, Food and Drug Administration Amendments Act; FDA, Food and Drug Administration; EMA, European Medicine Agency; PSUR, Periodic Safety Uptade Review; PMS, Post-Marketing Study; ADR, Adverse Drug Reaction.
- TOOLS
-
METRICS

-
- 0 Crossref
- Scopus
- 1,106 View
- 12 Download
-
Related articles in
J Korean Med Assoc -
Management and Surgical Treatment of Pressure Sore2002 September;45(9)
Diagnosis and Clinical Features of Food Poisoning2007 July;50(7)
Management of presbytinnitus2011 September;54(9)
Epidemiology, management, and prevention of cholera2017 February;60(2)