 |
 |
- Search
| J Korean Med Assoc > Volume 63(5); 2020 > Article |
|
Abstract
Since the first success of kidney transplantation in 1954, significant advances have been achieved in the field of organ transplantation. It was possible with the introduction of immunosuppressive drugs belonging to the class of calcineurin inhibitors (CNIs) such as cyclosporine and tacrolimus, the advances in surgical techniques and perioperative management, the monitoring and management infections, and the highly sensitive and specific antibody detection techniques. Despite recent progress, we currently encounter the limitation of better long-term transplant outcomes mainly because of paradoxical CNI toxicity and failure to control antibody or antibody-mediated rejections. The future direction of immunosuppression can be continued by optimizing immunosuppressive regimens with currently available immunosuppressants for better control of antibodies while avoiding CNI toxicity and by using biological therapeutics such as costimulation blockade agents that provide effective control of antibodies along with a reduction in usage or avoidance of CNIs and may develop as new immunosuppressants in the near future. Moreover, a tolerance induction through transplantation of donor hematopoietic stem cells or an infusion of regulatory cells using various sources of immune cells can also be a promising strategy as it can fundamentally escape from the complications of immunosuppressants. Over and above, it is important to note that the results of clinically applicable immunosuppressants from research in the non-human primate xenotransplantation model at the forefront of the future development of immunosuppressants can be a good opportunity to selectively apply to allo-transplants. No immunosuppressants can be risk-free, and therefore, all new immunosuppressants should be evaluated under the considerations for the risk/benefit ratio in various clinical conditions.
1954년에 첫 신장이식 성공, 1966년 췌장이식, 1967년 간이식의 성공이 이루어졌다. 이후 면역억제제인 싸이클로스포린(cyclosporine)과 타크로리무스(tacrolimis)의 개발 및 임상적용, 면역스크리닝 기법의 발전, 수술 술기의 발전 및 수술 전후의 환자관리의 경험축적에 따라 장기이식의 성적이 향상되면서, 장기이식은 말기장기부전 환자에서 궁극적인 치료법으로 자리잡았다.
최근에는 과거 이식의 금기사항으로 여겨졌던 혈액형 부적합 장기이식(ABO blood type incompatible trans-plantation)이 anti-CD20 antibody (rituximab), 혈장교환술 등의 조합으로 시행되는 전처치 프로토콜의 적용을 통해 이식 전에 혈액형에 대한 항체를 효과적으로 제거 또는 낮출 수 있게 되면서 보편화되기에 이르렀다. 또한 Luminex technique을 이용한 사람조직적합항원에 대한 항체의 검사법의 발전으로 민감하게 항체의 유무 및 세기의 평가가 가능하여, 면역학적으로 고위험 환자군인 고도 감작 환자에서 기증자 특이 항체를 스크리닝하여 이식 전 탈감작요법을 통한 장기이식을 시행하고, 이식 후에 항체의 변화를 모니터링하며 추적하고 있다.
장기이식 성적의 향상과 과거의 금기증을 극복한 적응증 확대(예: 혈액형 부적합 장기이식)에도 불구하고, 장기기증 수혜희망자 대비 기증장기의 부족은 더욱 심화되고 있고, 중·단기 이식성적의 향상에도 불구하고 장기(long-term)적인 장기이식의 성적의 향상은 한계에 봉착한 것처럼 보였다[1]. 장기적인 장기이식의 성적의 향상의 한계 원인으로는, 첫 번째로 현재 임상 장기이식의 주면역억제제인 싸이클로스포린, 타크로리무스의 칼시뉴린 억제제(calcineurin inhibitor, CNI) 신독성을 들 수 있다[2,3]. CNI로 인한 신독성은 신장이식이 아닌 심장, 간이식 환자에서도 신부전의 원인(이식 후 10년 만성신부전 약 20% 이상)이 되고 있고, 신장이식 환자에서 장기적인 면역억제제 복용 후에 거의 100%에서 신독성이 나타나서 이식장기의 기능이 저하되어 장기적인 이식편 성적 향상의 걸림돌로 여겨지고 있다. 두 번째로 장기이식 후 주요 사망원인으로 심혈관계질환, 감염, 악성질환의 발생을 들 수 있는데[4,5], 면역억제제 합병증으로 이식받은 환자들에게 다양한 종류의 기회 감염이 증가되며, 당뇨, 고혈압 등의 대사성질환의 발생 및 악화, 악성질환 발생의 위험증가에 기인한다 할 수 있다. 이식 환자에서 악성종양 발생률은 매우 다양한 암종에서 수 배에서 수십 배에 이르기까지 증가되며, 일반인에게서 흔히 나타나는 악성질환의 증가 외에도 헤르페스(herpes) type 8과 관련된 카포시 육종(Kaposi sarcoma)이나 엡스타인 바 바이러스(Epstein-Barr virus)와 관련된 비호치킨성 림프종이 이식 후 환자에서 빈발하는 대표적인 악성종양이다. 세 번째로, 장기적인 장기생존이 가능해지고, 민감한 검사법으로 항체 모니터링이 가능한 시대에, 이식 후 고위험군 환자에서 뿐 아니라 저위험군 환자에서도 새로운 기증자 특이 항체(de novo donor specific antibody, dnDSA) 발생으로 인한 항체 매개 거부반응(antibody-mediated rejection, AMR)이 이식편 기능부전의 주요 원인으로 알려지고 있다[6]. 임상적으로 이식편 기능이상이 나타나기 이전에 초·중반에 dnDSA발생이 선행하고, 현미경적 변화가 먼저 나타나는 것으로 이해되고 있는데[7], 현재의 면역억제제가 장기적인 경과에서 여전히 dnDSA발생 억제에 한계가 있거나 면역억제제 복용에 대한 환자 순응도 저하가 원인으로 이해되고 있다[6]. 여전히 거부반응을 예방하기 위한 효과적인 면역억제 조절과 면역억제제의 합병증의 최소화가 성공적인 장기이식에서 장기적인 성적 향상의 관건이라 할 수 있다.
익히 잘 알려진 CNI의 단점을 최소화하려는 노력은 현재도 활발히 진행되고 있는 접근이다. CNI를 주면역억제제로 하는 면역억제요법에서 CNI와 마이코페놀레이트 모페틸(mycophenolate mofetil) 또는 마이코페놀릭산(myco-phenolic acid)의 항대사제 또는 mammalian target of rapamycin (mTOR) 저해제와의 병합요법의 균형을 통해서 CNI의 부작용을 최소화하려는 요법은 현재 널리 받아들여지는 개념이다[8,9]. 그러나 장기간의 추적 결과에서 CNI 혈중 농도를 낮추는 것이 dnDSA의 발생을 높이는 위험이 있다고 알려져 이에 대한 재정립이 필요한 상황이다.
장기이식의 경험 축적과 아울러 재이식이 빈번해지고 있고, 과거 면역학적으로 금기증 또는 고위험군 외에도 면역학적 저위험군에서도 대부분의 면역억제제 임상시험에서 제시되는 단순한 단기의 거부반응 발생률의 비교 이상으로 장기적인 경과에서 dnDSA의 발생을 최소화하는가에 대한 다양한 비교연구들이 이루어지고 있다[10].
유지 면역억제제뿐 아니라 유도 면역억제제에서도 용량 감량과 dnDSA의 발생에서 장기적인 효과에 대한 임상연구들이 지속되고 있다. 유도 면역억제제 중 전반적인 T세포를T-cell depleting하는 항 흉선세포 글로불린을 투여한 환자군에서 dnDSA의 발생이 적었다고 보고하였으나[11] 다양한 환자군에 비교분석이 필요하다.
CNI 면역억제제는 합병증으로 인해 혈중 농도의 모니터링 하에서 복용해야 하는 약제로, 환자내에서의 면역억제제 혈중 농도 변이(intrapatient variability, IPV)가 장기 이식편 기능부전에 영향을 미치는 인자 중 하나로 제시되면서, 다양한 환자군에서 면역억제제의 IPV가 장기적인 이식편 생존율에 영향을 미치는지에 대한 임상 데이터들이 축적되어 비교평가 되는 과정에 있다[12,13]. IPV는 현실의 임상 적용 영역에서 환자의 순응도 외에도 면역억제제의 흡수율의 변화 등의 다양한 요인의 영향을 받을 수 있기에 IPV를 임상적용의 영역에서 환자의 순응도를 높이기 위해 활용될 것인지에 대한 추가적인 연구가 필요하다[14].
현재는 과거의 부진한 이식 성적 속에서 싸이클로스포린, 타크로리무스와 같은 효과적인 새로운 면역억제의 출현으로 획기적인 성적 향상을 기대하기 어려운 시대에 있다고 생각된다. 장기적인 이식 성적의 향상은 다양한 면역학적인 위험요소를 안고 있는 환자에서 단순히 하나의 면역억제제 또는 면역억제 요법을 선택하는가의 문제만은 아니다. 현재 임상에 적용 가능한 면역억제제 중에서 민감한 항체 모니터링을 통해 장기적인 dnDSA 발생을 효과적으로 억제할 수 있는 조합의 면역억제요법이 필요하다. 또한 이식 시기에 따른 면역억제요법의 전환 과정에서 환자의 순응도를 포함하여 면역억제제 혈중 농도를 안정적으로 유지하는 등의 총합의 결과로 달성될 수 있을 것이다. 이에 기존의 면역억제제 하에도 기존의 항체 모니터링을 포함하는 최적화의 모니터링 프로토콜의 정립이 장기적인 이식성적 향상을 위한 방향이라 할 수 있다.
CNI의 합병증을 대체할 수 있을 것으로 기대되는 면역억제제의 주된 목표로 공동자극경로를 들 수 있다. 외부 항원에 의한 T세포 활성화는 T세포 수용체에 의한 신호전달(signal 1) 이후 공동자극(signal 2) 및 사이토카인 자극(signal 3)에 의해 이루어지는데, 이들 세 가지 신호 중에서 공동자극에 의한 자극은 T세포 활성화에 매우 중요하며, 이 신호가 억제될 경우 T세포가 외부 항원에 무반응 상태에 빠지는 것으로 알려져 있고, 이식면역에 있어서 장기이식 후 면역반응 조절을 위한 핵심 타겟이다[15,16]. 공동자극경로는 최근 항암치료에서 면역관문으로도 불리며, 면역치료 분야에서 각광받는 주요 타겟이기도 하다. 이때 면역치료에서는 T세포의 면역을 증강시키는 방향으로 치료에 적응되나, 이식면역에서는 T세포의 면역을 억제하는 방향으로 적용하고자 한다는 점이 다른 점이라 할 수 있다. 이식면역에서는 signal 2의 공동자극경로 중에서는 CD80,86/CD28, CD40/CD40L (CD154) pathway 등이, 공동저해경로로는 CD80,86/cytotoxic T lymphocyte antigen (CTLA)-4, programmed death (PD)-1/PD-L1,2 pathway가 주요한 개발 타겟으로 연구되고 있다(Figure 1).
현재 미국 식품의약국 승인을 받고 임상에서 사용 가능한 공동자극경로 억제제로 TLA4-Ig인 belatacept가 있다[17]. Emory 대학 Lowe 등[18]과 Kirk 등[19]은 CNI 대체제로 belatacept와 mTOR 억제제인 sirolimus를 사용하여 원숭이 동종 신장 및 췌도 이식에서의 효능을 검증함으로써, 공동자극인자 억제제를 CNI 대체제로서의 가능성을 보여주었고, belatacept 효과에 대해 전임상 영장류 감작 모델에서 탈감작 후 림프절의 배중심에서의 항체 생성의 체액성 반등을 예방하는 것으로 보고하였으며, 임상에서는 이전 연구(BENEFIT study)의 연장연구에서 belatacept 투여군에서 이식 후에 항체 생성을 억제하는 것으로 보고하였다[20,21]. Belatacept가 T세포뿐 아니라 B세포, 항체 생성의 억제에도 역할이 있는 것으로 보고되고 있어, CNI의 대체제 뿐 아니라 항체 생성 억제 측면에서 향후에도 여전히 역할이 기대되고 있다.
또 하나의 가능성이 있는 공동자극경로(CD40/CD40L[CD154] pathway) 저해를 통한 면역억제제가 항CD154(CD40L) 항체(anti-CD154 Ab)이다. 전임상 영장류 실험에서 항CD154 항체의 T세포 조절기능이 우수함이 다양한 이식실험모델에서 증명된 바 있으나, 자가면역질환(루푸스신장염)에서 임상시험 중 항체의 Fc portion의 일부가 혈소판에 부착하여 혈전색전증의 부작용이 발생되어 임상시험이 중단된 바 있다[22]. 이러한 부작용에도 불구하고 다양한 이식모델의 전임상 영장류 동종 및 이종 이식 연구에서 항CD154 항체의 사용이 CTLA-4 Ig 혹은 항CD40 항체에 비하여 효과적임이 보고되고 있고, 특히 현재까지 다양한 유전자조작 돼지를 이용한 영장류 이종이식 모델에서 주된 면역억제제로 좋은 성적을 보이고 있어, 과거 혈전색전증에 대한 부작용을 극복, 조절한 신약으로 면역질환 및 이식에서의 면역억제제로서 연구개발이 진행되고 있다[23,24].
최근에는 CD40/CD40L (CD154)pathway를 타겟으로 하는 항CD40 항체인 iscalimab (CFZ533)이 신장이식 환자들을 대상으로 CNI 대체 면역억제제로서의 가능성에 대한 임상 시도(phase 2b) 중에 있으며(NCT03663335) [25] 또 다른 항CD40 항체인 bleselumab(ASKP1240)이 신장이식에서 임상시험 중에 있다(NCT01780844) [26].
항체의 생성 및 항체 매개의 면역반응은 장기이식에서 면역학적으로 이식편 부전의 주요 요인인데, 공동자극 억제제의 체액성 면역반응에서 효과적인 억제 작용이 있음이 잘 알려져 있어[27], belatacept 이후에 공동자극 억제제가 동종이식 및 이종이식모델을 기반으로 면역억제제로서 집중적으로 연구개발되고 있다. 이러한 공동자극 억제제는 앞서 기존의 mTOR 저해제 등의 면역억제제와의 조합에서 항체 생성을 억제하는 역할에 대한 연구도 이루어지고 있다.
유지 면역억제제로서의 역할은 아니지만 제한되나마 신장이식 후 탈감작요법과 AMR의 치료에서 다양한 면역억제제가 사용 또는 시도되고 있다. 앞서 전처치 또는 탈감작요법에서 rituximab (항CD20 ab)의 사용에 대해 기술하였다. Rituximab은 pan B-cell의 marker인 CD20에 대한 항체로 림프종의 주요한 치료제로 쓰여 왔다. 항체를 생성하는 형질세포에 특이적인 항체는 아니지만 항체를 만들어내는 B세포를 전반적으로 감소시킴으로써 궁극적으로 항체를 조절하는 작용으로 전처치 또는 탈감작요법에서 장기이식의 영역에서 사용되어 왔다. 그 외에 항체를 생성하는 형질세포 타겟의 proteosome inhibitor (bortezomib)도 제한적으로 사용되고 있으나 초기의 기대에는 못 미치는 임상결과를 보고하고 있다. 그러나 차세대 항CD20 ab인 obinutuzumab [28], 차세대 proteosome inhibitor인 carfilzomib이 연구되고 있으며[29], 그 외에도 보체경로 타겟의 C5 inhibitor (eculizumab)와 anti-IL-6 receptor Ab (tocilizumab), 항BAFF 항체 등 다양한 항체 형성 억제 타겟으로 하는 생물학적 제제들이 연구개발 되고 있다.
현재로는 절대적인 치료효과가 입증되어 널리 임상 적용되는 AMR치료제는 명확치 않으나 향후 AMR 또는 만성 활동성 AMR에 대한 지속적인 시도가 이루어지고 있고, 그에 대한 축적된 임상결과는 우리가 추구하는 장기적인 장기이식 성적 향상에 기여할 것으로 기대된다.
면역억제제의 합병증과 부작용으로부터 벗어나 장기적인 장기(allograft) 생존 향상을 달성하고자 하는 이상적인 미래의 면역억제 전략 중의 하나가 면역억제제를 유지하지 않고도 안정된 이식편 기능을 유지하게 하는 면역 관용의 유도이다. 많은 연구자들은 효과적인 면역억제제가 부재하였던 시대 이래로 면역억제제 없이도 안정된 이식편 기능 유지를 꿈꾸어 왔다. 이식면역 연구와 임상 장기이식의 발전 역사 속에서 기증자와 수혜자의 면역체계가 공존하는 혼합 카이메리즘(mixed chimerism)의 현상이 면역 관용 유도의 한 방법으로 전임상의 과정을 통해 제시되어왔다. 임상에서의 첫 결과로 2008년 Massachusetts General Hospital의 Kawai 등[30]이 골수와 신장을 동시에 이식받은 다섯 명의 환자 중 네 명에서 신장이식 후 면역억제제를 끊는데 성공하였다. 이 연구에서 이식 환자에서 일시적인 혼합 카이메리즘이 유도되어 이식 후 수여자에서 면역관용이 유도되는 장기간의 결과를 보여주었다. 국내에서도 전처치 후 신장, 골수 동시 이식(combined kidney and bone marrow transplantation)을 통한 성공적인 면역관용 유도가 보고되었다[31]. 한편, Northwestern Memorial Hospital의 Leventhal 등[32]은 기증자의 말초 조혈모세포로부터 분리한, 항원 특이적인 조절 T세포(regulatory T-cell, Treg)의 생성을 유도한다는 “facilitating cell”을 신장이식과 동시에 시행하면서 지속적인, 견고한 카이메리즘을 통해 장기적인 면역관용 유도에 성공하였다. 특히 그의 연구는 면역억제제를 개발하는 다국적 제약회사의 주도로 facilitating cell therapy로 임상시도가 진행되고 있다(NCT03995901). 면역관용의 유도는 현재 몇개의 센터에서 제한된 환자에서 진행되는 임상 시도이지만, 면역억제제의 합병증으로부터 자유로워질 수 있다는 점에서 면역억제제의 장기 사용으로 인한 이식편 기능부전 없이 “평생에 한 번의 이식”을 목표로 가까운 미래에 궁극의 면역억제 전략으로 많은 환자들에게 적용되기를 기대해 본다.
면역관용을 목표로 하는 주요 시도 중의 하나가 Treg cell을 이용한 치료이다. Todo 등[33]은 생체 간이식 pilot study에서 10명의 환자 중 체외에서 생성된 Treg cell (ex vivo-generated regulatory T-cell-enriched cell) 투여를 통해서 일곱 명의 환자에서 성공적으로 면역관용을 유도하였다. 한편 임상 신장이식 환자들을 대상으로 전 세계적인 다기관 연구(the ONE study consortium)에서 polyclonal regulatory T-cell, donor alloantigen reactive regulatory T-cell, regulatory donor derived macrophage, dendritic cell 등 Treg cell을 포함한 다양한 세포를 이용한 면역관용 유도를 위한 안전성 및 적용 가능성에 대한 임상연구가 현재 진행 중에 있다[34]. 기타 다른 세포치료로는 면역조절기능이 있다고 알려진 중배엽 줄기세포를 이용한 시도들이 Tan 등[35]의 임상시험 이래 많은 임상시험이 현재 진행 중에 있다.
이종이식은 서론에 기술한 현재의 장기이식의 한계인 기증장기 부족을 극복할 수 있는 무한한 장기공급의 원천적인 해결책이다[36]. 미래의 면역억제제에서 이종이식의 전임상연구 결과를 주목해야 이유는 현재 장기이식의 한계인 기증장기 부족의 문제를 해결해 줄 뿐만 아니라 임상 동종이식이식에서 항체 생성 억제 및 항체매개 거부반응의 치료의 해결책을 제시해줄 수 있기 때문이다. 이종이식은 앞서 언급한 수많은 면역억제 전략을 총동원하여 전임상 영장류 실험을 기반으로 임상진입을 목표로 하고 있다. 특히 최근 CRSPR Cas9의 기술이 형질전환 돼지의 개발에 적용되어, 다중유전자 knock-out, knock-in이 가능하고 빠른 기간(수 개월) 내에 형질전환 돼지가 생산되어 검증 가능하게 되는 등 과거 약 20년간의 성과 이상의 성과가 최근 2-3년 사이에 이루어졌다. 최근에는 열 개 이상의 유전자 편집된 돼지가 만들어지고 있다. 특히 전임상 영장류 시험결과도 획기적으로 향상되어, 이종 신장이식에서 α1,3-galactosyltransferase (GalT) knock-out, 인간 CD55발현 돼지를 이용하여 이종 신장이식 모델에서 499일의 이식편 생존을 보고하였다[37]. 한편 영장류 이종심장이식 시험 분야에서는 GalT 제거(knock-out), 인간 막 보조인자 단백(human membrane cofactor protein, CD46)과 인간 트롬보모둘린(human thrombomodulin)이 발현하는 돼지를 이용한 정위적 이종 심장이식에서 195일의 이식편 생존율이 보고되었다[38]. Langin 등[38]의 성과가 주목받는 이유는 과거의 945일의 이식편 생존의 기록이 이소성 이종심장 이식편이 단지 생존한 결과와 달리 순환기능을 완전히 대체하는 정위적 이종심장의 결과라는 점에 있다. 과거에 생산된 제한된 유전자 편집된 돼지를 이용한 영장류 이종이식 모델에서의 결과라는 점에서 가까운 미래에 다중유전자 편집 돼지에서의 더 좋은 결과가 기대된다.
이러한 전임상 영장류 이종이식 모델에서 사용되는 면역억제제가 바로 anti-CD20, anti-CD4, anti-CD8, anti-thymocyte globulin, anti-CD154, anti-CD40, CTLA4-Ig 등의 조합이다. 이종이식의 임상진입단계에서의 걸림돌 중의 하나가 임상적용이 가능한 이종이식에서 효과적인 면역억제 요법이다. 현재 동종이식에서 임상적용 가능한 면역억제제 이상의 면역억제제가 이종이식에서 요구되는데, 이러한 면역억제제 중 anti-CD40 등 일부는 임상적용 시도들이 이미 진행되고 있고, anti-CD154 등은 임상시험 전단계의 전임상 영장류 실험 등이 활발히 이루어지고 있다는 점에서 이종이식의 전임상 실험연구가 동종이식의 면역억제제 개발의 최전선 역할을 하고 있다고 생각된다. 물론 이종이식에서 효과적인 면역억제제가 동종이식에서 유용한가에 대한 고민은 감염의 위험, 악성질환의 발생률 증가 등 위험 대비 이득의 관점에서 연구개발 뒤에 진지하게 고민해야 할 것이다.
현재의 장기이식은 CNI의 효과에 힘입어 면역학적으로 저위험군 환자에서 합격점 이상의 성적을 나타내고 있다고 생각한다. 그러나 장기적인 이식성적의 향상 또는 특히 고위험군 환자에서의 향상된 이식 성적을 위해서는 신독성으로 대표되는CNI 합병증을 피하면서도 효과적으로 항체 및 AMR을 조절하기위한 미래의 면역억제 요법의 전략이 필요하다(Figure 2).
첫째, CNI의 신독성을 피하면서도 민감한 항체 모니터링을 통해 기존의 면역억제제의 조합들 중 최적화 면역억제요법을 정립하고, 둘째로 공동자극 억제제 등의 생물학제제는 CNI를 능가하고 부작용을 줄이며, 효과적으로 항체를 억제할 수 있는 가까운 미래의 새로운 면역억제가 될 수 있을 것이다. 셋째로 조혈모세포이식 또는 다양한 근원의 면역조절 세포를 이용한 면역관용 유도 역시 면역억제제의 합병증으로부터 근본적으로 벗어날 수 있는 전략이 되겠다. 넷째로 면역억제제 개발의 최전선에 있는 이종이식모델에서의 다양한 결과로부터 임상적으로 적용 가능한 면역억제제의 결과들이 동종이식에 선택적으로 적용될 수 있는 좋은 기회가 될 수 있기에 주목해야 하겠다. 어떤 새로운 면역억제제도 감염 등 합병증의 위험으로부터 자유로울 수는 없기에 개개 환자에서 위험 대비 이득에 대한 냉철한 평가를 통해 적용해야 할 것으로 여겨진다.
Figure 1.
Co-stimulation (signal 2) for the target of immune suppression. CD, cluster of differentiation, CTLA, cytotoxic T lymphocyte antigen; PD, programmed death; DC, dendritic cell; ICOS, inducible T-cell costimulatory; ICAM, intercellular adhesion molecule; LFA, lymphocyte function-associated antigen.
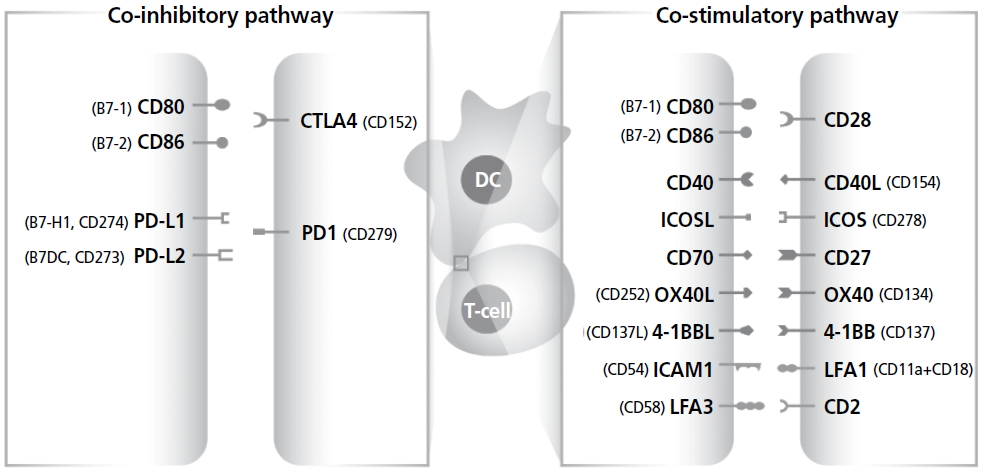
Figure 2.
Future direction of immune suppression in organ transplantation. IS, immune suppression; BM, bone marrow; HSC, hematopoietic stem cell; CNI, calcineurin inhibitor; dnDSA, de novo DSA; DSA, donor specific antibody; R&D, research and development; NHP, nonhuman primate; TG, transgeneic; DFP, designated pathogen-free.

References
1. Lodhi SA, Lamb KE, Meier-Kriesche HU. Solid organ allograft survival improvement in the United States: the long-term does not mirror the dramatic short-term success. Am J Transplant 2011;11:1226-1235.


2. Ojo AO, Held PJ, Port FK, Wolfe RA, Leichtman AB, Young EW, Arndorfer J, Christensen L, Merion RM. Chronic renal failure after transplantation of a nonrenal organ. N Engl J Med 2003;349:931-940.


3. Nankivell BJ, Borrows RJ, Fung CL, O'Connell PJ, Allen RD, Chapman JR. The natural history of chronic allograft nephropathy. N Engl J Med 2003;349:2326-233.


4. Pascual M, Theruvath T, Kawai T, Tolkoff-Rubin N, Cosimi AB. Strategies to improve long-term outcomes after renal transplantation. N Engl J Med 2002;346:580-590.


5. van Leeuwen MT, Webster AC, McCredie MR, Stewart JH, McDonald SP, Amin J, Kaldor JM, Chapman JR, Vajdic CM, Grulich AE. Effect of reduced immunosuppression after kid-ney transplant failure on risk of cancer: population based retrospective cohort study. BMJ 2010;340:c570.



6. Sellares J, de Freitas DG, Mengel M, Reeve J, Einecke G, Sis B, Hidalgo LG, Famulski K, Matas A, Halloran PF. Under-standing the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am J Transplant 2012;12:388-399.


7. Colvin RB, Smith RN. Antibody-mediated organ-allograft rejection. Nat Rev Immunol 2005;5:807-817.



8. Ekberg H, Tedesco-Silva H, Demirbas A, Vitko S, Nashan B, Gurkan A, Margreiter R, Hugo C, Grinyo JM, Frei U, Van-renterghem Y, Daloze P, Halloran PF; ELITE-Symphony Study. Reduced exposure to calcineurin inhibitors in renal transplantation. N Engl J Med 2007;357:2562-2575.


9. Kreis H, Oberbauer R, Campistol JM, Mathew T, Daloze P, Schena FP, Burke JT, Brault Y, Gioud-Paquet M, Scarola JA, Neylan JF; Rapamune Maintenance Regimen Trial. Long-term benefits with sirolimus-based therapy after early cyclosporine withdrawal. J Am Soc Nephrol 2004;15:809-817.


10. Senev A, Lerut E, Van Sandt V, Coemans M, Callemeyn J, Sprangers B, Kuypers D, Emonds MP, Naesens M. Specificity, strength, and evolution of pretransplant donor-specific HLA antibodies determine outcome after kidney transplantation. Am J Transplant 2019;19:3100-3113.


11. Brokhof MM, Sollinger HW, Hager DR, Muth BL, Pirsch JD, Fernandez LA, Bellingham JM, Mezrich JD, Foley DP, D'Alessandro AM, Odorico JS, Mohamed MA, Vidyasagar V, Ellis TM, Kaufman DB, Djamali A. Antithymocyte globulin is associated with a lower incidence of de novo donor-specific antibodies in moderately sensitized renal transplant recipients. Transplantation 2014;97:612-617.



12. Shuker N, Shuker L, van Rosmalen J, Roodnat JI, Borra LC, Weimar W, Hesselink DA, van Gelder T. A high intrapatient variability in tacrolimus exposure is associated with poor long-term outcome of kidney transplantation. Transpl Int 2016;29:1158-1167.


13. Sablik KA, Clahsen-van Groningen MC, Hesselink DA, van Gelder T, Betjes MGH. Tacrolimus intra-patient variability is not associated with chronic active antibody mediated rejec-tion. PLoS One 2018;13:e0196552.



14. Leino AD, King EC, Jiang W, Vinks AA, Klawitter J, Christians U, Woodle ES, Alloway RR, Rohan JM. Assessment of tacro-limus intrapatient variability in stable adherent transplant recipients: establishing baseline values. Am J Transplant 2019;19:1410-1420.


15. Jenkins MK, Schwartz RH. Antigen presentation by che-mically modified splenocytes induces antigen-specific T cell unresponsiveness in vitro and in vivo. J Exp Med 1987;165:302-319.




16. Halloran PF. Immunosuppressive drugs for kidney trans-plantation. N Engl J Med 2004;351:2715-2729.


17. Rostaing L, Vincenti F, Grinyo J, Rice KM, Bresnahan B, Steinberg S, Gang S, Gaite LE, Moal MC, Mondragon-Ram-irez GA, Kothari J, Pupim L, Larsen CP. Long-term belatacept exposure maintains efficacy and safety at 5 years: results from the long-term extension of the BENEFIT study. Am J Transplant 2013;13:2875-2883.


18. Lowe MC, Badell IR, Turner AP, Thompson PW, Leopardi FV, Strobert EA, Larsen CP, Kirk AD. Belatacept and sirolimus prolong nonhuman primate islet allograft survival: adverse consequences of concomitant alefacept therapy. Am J Trans-plant 2013;13:312-319.

19. Kirk AD, Guasch A, Xu H, Cheeseman J, Mead SI, Ghali A, Mehta AK, Wu D, Gebel H, Bray R, Horan J, Kean LS, Larsen CP, Pearson TC. Renal transplantation using belatacept without maintenance steroids or calcineurin inhibitors. Am J Transplant 2014;14:1142-1151.



20. Kim EJ, Kwun J, Gibby AC, Hong JJ, Farris AB 3rd, Iwakoshi NN, Villinger F, Kirk AD, Knechtle SJ. Costimulation bloc-kade alters germinal center responses and prevents antibody-mediated rejection. Am J Transplant 2014;14:59-69.



21. Bray RA, Gebel HM, Townsend R, Roberts ME, Polinsky M, Yang L, Meier-Kriesche HU, Larsen CP. De novo donor-specific antibodies in belatacept-treated vs cyclosporine-treated kidney-transplant recipients: post hoc analyses of the randomized phase III BENEFIT and BENEFIT-EXT studies. Am J Transplant 2018;18:1783-1789.



22. Kawai T, Andrews D, Colvin RB, Sachs DH, Cosimi AB. Thromboembolic complications after treatment with mono-clonal antibody against CD40 ligand. Nat Med 2000;6:114.


23. Karnell JL, Rieder SA, Ettinger R, Kolbeck R. Targeting the CD40-CD40L pathway in autoimmune diseases: Humoral immunity and beyond. Adv Drug Deliv Rev 2019;141:92-103.


24. Shock A, Burkly L, Wakefield I, Peters C, Garber E, Ferrant J, Taylor FR, Su L, Hsu YM, Hutto D, Amirkhosravi A, Meyer T, Francis J, Malcolm S, Robinson M, Brown D, Shaw S, Foulkes R, Lawson A, Harari O, Bourne T, Maloney A, Weir N. CDP7657, an anti-CD40L antibody lacking an Fc domain, inhibits CD40L-dependent immune responses without thro-mbotic complications: an in vivo study. Arthritis Res Ther 2015;17:234.



25. Vincenti F, Klintmalm G, Yang H, Ram Peddi V, Blahunka P, Conkle A, Santos V, Holman J. A randomized, phase 1b study of the pharmacokinetics, pharmacodynamics, safety, and tolerability of bleselumab, a fully human, anti-CD40 monoclonal antibody, in kidney transplantation. Am J Trans-plant 2020;20:172-180.

26. Harland RC, Klintmalm G, Jensik S, Yang H, Bromberg J, Holman J, Kumar MSA, Santos V, Larson TJ, Wang X. Efficacy and safety of bleselumab in kidney transplant recipients: a phase 2, randomized, open-label, noninferiority study. Am J Transplant 2020;20:159-171.


27. Leibler C, Thiolat A, Elsner RA, El Karoui K, Samson C, Grimbert P. Costimulatory blockade molecules and B-cell-mediated immune response: current knowledge and perspec-tives. Kidney Int 2019;95:774-786.


28. Choi J, Vo A, Huang E, Louie S, Kang A, Peng A, Najjar R, Jordan S. Experience with Obinutuzumab (Type II Anti-CD20) in kidney transplant patients with donor specific anti-body (DSA+) antibody mediated rejection [abstract]. Am J Transplant 2017;17 Suppl 3:310.
29. Tremblay S, Driscoll JJ, Rike-Shields A, Hildeman DA, Allo-way RR, Girnita AL, Brailey PA, Woodle ES. A prospective, iterative, adaptive trial of carfilzomib-based desensitization. Am J Transplant 2020;20:411-421.


30. Kawai T, Cosimi AB, Spitzer TR, Tolkoff-Rubin N, Suthan-thiran M, Saidman SL, Shaffer J, Preffer FI, Ding R, Sharma V, Fishman JA, Dey B, Ko DS, Hertl M, Goes NB, Wong W, Williams WW Jr, Colvin RB, Sykes M, Sachs DH. HLA-mismatched renal transplantation without maintenance immunosuppression. N Engl J Med 2008;358:353-361.



31. Lee KW, Park JB, Park H, Kwon Y, Lee JS, Kim KS, Chung YJ, Rhu JS, Choi S, Kwon GY, Kim HJ, Kang ES, Jung CW, Shin EC, Kawai T, Kim SJ, Joh JW. Inducing transient mixed chimerism for allograft survival without maintenance immunosuppression with combined kidney and bone marrow transplantation: protocol optimization. Transplantation 2019;Oct. 18. [Epub]. https://doi.org/10.1097/TP.0000000000003006

32. Leventhal J, Abecassis M, Miller J, Gallon L, Ravindra K, Tollerud DJ, King B, Elliott MJ, Herzig G, Herzig R, Ildstad ST. Chimerism and tolerance without GVHD or engraftment syndrome in HLA-mismatched combined kidney and hematopoietic stem cell transplantation. Sci Transl Med 2012;4:124. ra28.

33. Todo S, Yamashita K, Goto R, Zaitsu M, Nagatsu A, Oura T, Watanabe M, Aoyagi T, Suzuki T, Shimamura T, Kamiyama T, Sato N, Sugita J, Hatanaka K, Bashuda H, Habu S, Demetris AJ, Okumura K. A pilot study of operational tolerance with a regulatory T-cell-based cell therapy in living donor liver trans-plantation. Hepatology 2016;64:632-643.


34. Kawai T, Leventhal J, Wood K, Strober S. Summary of the Third International Workshop on Clinical Tolerance. Am J Transplant 2019;19:324-330.


35. Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, Sun X, Chen J, Yang S, Cai J, Gao X, Pileggi A, Ricordi C. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA 2012;307:1169-1177.


36. Park CG, Kim JS, Kim YH. Current status and future pers-pectives of xenotransplantation and stem cell research in transplantation field. J Korean Med Assoc 2008;51:732-744.

37. Kim SC, Mathews DV, Breeden CP, Higginbotham LB, Ladowski J, Martens G, Stephenson A, Farris AB, Strobert EA, Jenkins J, Walters EM, Larsen CP, Tector M, Tector AJ, Adams AB. Long-term survival of pig-to-rhesus macaque renal xenografts is dependent on CD4 T cell depletion. Am J Transplant 2019;19:2174-2185.



38. Langin M, Mayr T, Reichart B, Michel S, Buchholz S, Guethoff S, Dashkevich A, Baehr A, Egerer S, Bauer A, Mihalj M, Panelli A, Issl L, Ying J, Fresch AK, Buttgereit I, Mokelke M, Radan J, Werner F, Lutzmann I, Steen S, Sjoberg T, Paskevicius A, Qiuming L, Sfriso R, Rieben R, Dahlhoff M, Kessler B, Kemter E, Kurome M, Zakhartchenko V, Klett K, Hinkel R, Kupatt C, Falkenau A, Reu S, Ellgass R, Herzog R, Binder U, Wich G, Skerra A, Ayares D, Kind A, Schonmann U, Kaup FJ, Hagl C, Wolf E, Klymiuk N, Brenner P, Abicht JM. Consistent success in life-supporting porcine cardiac xeno-transplantation. Nature 2018;564:430-433.



Peer Reviewers’ Commentary
이 논문은 장기이식 면역억제요법의 과거 및 현재 동향과 함께, 동종이식, 면역 관용, 이종이식에서 사용 가능한 면역억제제와 미래 가능한 면역억제요법에 관한 최신 지식을 정리한 논문이다. 면역억제요법의 궁극적인 목표는 면역학적으로는 거부반응, 특히 항체 매개 거부반응 발생을 줄이고, 면역억제제 사용에 따른 감염 및 악성종양 발생과 같은 면역억제 관련 합병증을 최소화하여 환자 및 이식편의 생존율을 높이는 것이다. 이를 위하여 여러 가지 면역억제제들이 개발되고 있으며 이러한 면역억제제들을 환자의 면역학적 위험도를 고려해 적절히 조합하여 사용하고 공여자 특이 항체 발생을 모니터링하는 것이 중요하다. 이 논문은 최신 면역억제 요법과 함께 앞으로의 면역억제 요법의 방향을 제시해 주고 있어 장기이식 관련 임상 현장에 많은 도움을 줄 것으로 판단된다.
[정리: 편집위원회]
-
METRICS

-
- 1 Crossref
- Scopus
- 5,528 View
- 283 Download
-
Related articles in
J Korean Med Assoc -
Direction of diagnosis and treatment improvement in colorectal cancer2022 September;65(9)
Mechanisms and clinical applications of immunosuppressive medications2020 May;63(5)
Brain Death Donor Organ Transplantation1999 April;42(4)
Recent Advancement in Renal Transplantation2003 October;46(10)
Future Prospect of the Silver Industry in Korea and Physicians' Participation2008 November;51(11)






