|
 |
- Search
| J Korean Med Assoc > Volume 55(8); 2012 > Article |
Abstract
Around the world electronic health records data are being shared and exchanged between two different systems for direct patient care, as well as for research, reimbursement, quality assurance, epidemiology, public health, and policy development. It is important to communicate the semantic meaning of the clinical data when exchanging electronic health records data. In order to achieve semantic interoperability of clinical data, it is important not only to specify clinical entries and documents and the structure of data in electronic health records, but also to use clinical terminology to describe clinical data. There are three types of clinical terminology: interface terminology to support a user-friendly structured data entry; reference terminology to store, retrieve, and analyze clinical data; and classification to aggregate clinical data for secondary use. In order to use electronic health records data in an efficient way, healthcare providers first need to record clinical content using a systematic and controlled interface terminology, then clinical content needs to be stored with reference terminology in a clinical data repository or data warehouse, and finally, the clinical content can be converted into a classification for reimbursement and statistical reporting. For electronic health records data collected at the point of care to be used for secondary purposes, it is necessary to map reference terminology with interface terminology and classification. It is necessary to adopt clinical terminology in electronic health records systems to ensure a high level of semantic interoperability.
Acknowledgement
This research was supported by the basic science research program through the National Research Foundation of Korea funded by the Ministry of Education, Science and Technology (2010-0028631). YRP's education grant was supported by the Ministry of Health and Welfare, Republic of Korea (A112020).
References
1. Dolin RH, Alschuler L, Boyer S, Beebe C, Behlen FM, Biron PV, Shabo Shvo A. HL7 clinical document architecture, release 2. J Am Med Inform Assoc 2006;13:30-39.
2. American Society for Testing and Materials. Standard specification for continuity of care record (CCR) [Internet] 2005;cited 2012 Jun 28. West Conshohocken: ASTM International. Available from: http://www.astm.org/Standards/E2369.htm
3. Clinical Data Interchange Standards Consortium. Operational data model [Internet] 2012;cited 2012 Jun 28. Round Rock: Clinical Data Interchange Standards Consortium. Available from: http://www.cdisc.org/odm
4. Hurrell MJ, Monk TG, Nicol A, Norton AN, Reich DL, Walsh JL. Implementation of a standards-based anaesthesia record compliant with the health level 7 (HL7) clinical document architecture (CDA). J Clin Monit Comput 2012;26:295-304.
5. Otter-Nickerson B. HIE: the interoperable way to deliver quality healthcare: finally, the concept of health information exchange is being more concretely defined and accepted by the healthcare community. Health Manag Technol 2011;32:19.
6. Richesson RL, Krischer J. Data standards in clinical research: gaps, overlaps, challenges and future directions. J Am Med Inform Assoc 2007;14:687-696.
7. Smith B, Ceusters W. HL7 RIM: an incoherent standard. Stud Health Technol Inform 2006;124:133-138.
8. Humphreys BL, Lindberg DA, Schoolman HM, Barnett GO. The unified medical language system: an informatics research collaboration. J Am Med Inform Assoc 1998;5:1-11.
9. Cote RA, Rothwell DJ, Palotay JL, Beckett RS, Brochu L. College of American Pathologists. American Veterinary Medical Association. The systematized nomenclature of human and veterinary medicine: SNOMED international 1993;3rd ed. Northfield: College of American Pathologists.
10. Solbrig HR. Metadata and the reintegration of clinical information: ISO 11179. MD Comput 2000;17:25-28.
11. Agency for Healthcare Research and Quality. United States Health Information Knowledgebase [Internet] 2012;cited 2012 Jun 18. Rockville: Agency for Healthcare Research and Quality. Available from: http://ushik.ahrq.gov
12. Warzel DB, Andonaydis C, McCurry B, Chilukuri R, Ishmukhamedov S, Covitz P. Common data element (CDE) management and deployment in clinical trials. AMIA Annu Symp Proc 2003;1048.
13. International Organization for Standardization. International Electrotechnical Commission. Information technology: specification and standardization of data elements. Part 3. Basic attributes of data elements 1994;Geneva: ISO/IEC 11179-3.
14. Fridsma DB, Evans J, Hastak S, Mead CN. The BRIDG project: a technical report. J Am Med Inform Assoc 2008;15:130-137.
15. Buetow KH, Niederhuber J. Infrastructure for a learning health care system: CaBIG. Health Aff (Millwood) 2009;28:923-924.
16. Costa CM, Menarguez-Tortosa M, Fernandez-Breis JT. Clinical data interoperability based on archetype transformation. J Biomed Inform 2011;44:869-880.
17. Clinical Data Interchange Standards Consortium. Clinical data acquisition standards harmonization [Internet] 2012;cited 2012 Jun 28. Round Rock: Clinical Data Interchange Standards Consortium. Available from: http://www.cdisc.org/cdash
Figure 1
Creating metadata for clinical document exchange. (A) Basic architecture of ISO/IEC 11179 consists of data element, data element concept (DEC), conceptual domain and value domain. (B) Procedure diagram of metadata extraction vocabulary mapping from clinical documents. UMLS, unified medical language system.
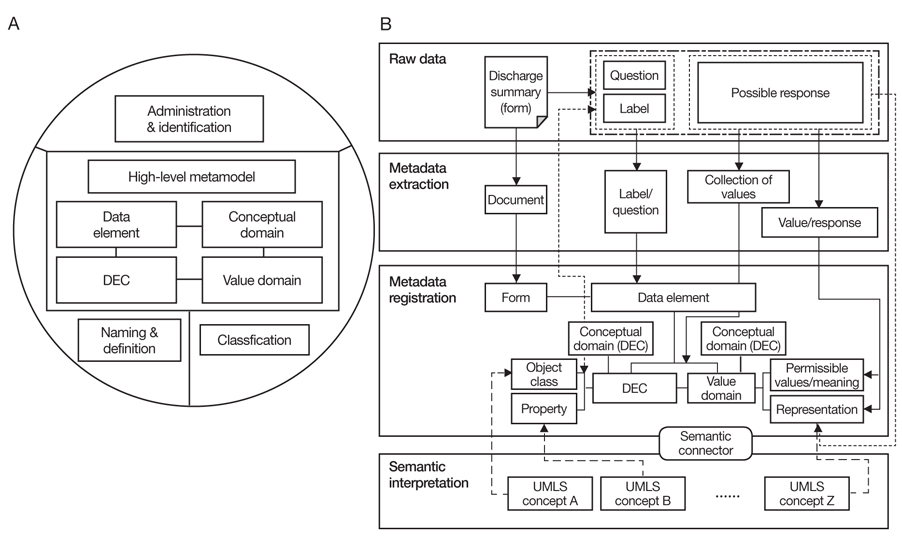
Figure 2
The example of medadata about patient personal medical history asthma occurrence indicator.
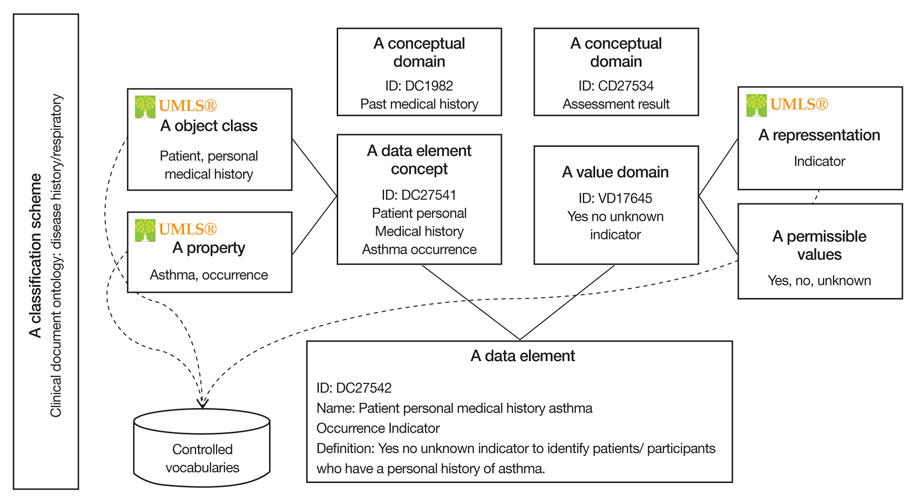
Figure 3
Metadata registry (MDR)-based semantic interoperability during clinical document architecture (CDA)-based clinical document exchange. EMR, electronic medical record; OpenEHR, open Electronic Health Record.
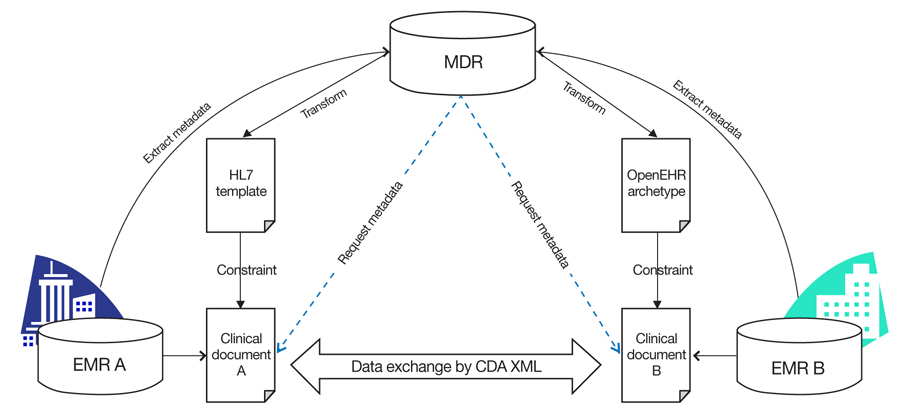
Figure 4
Example for (A) HL-7 template and (B) open Electronic Health Record archetype on patient registration number.

Table 1
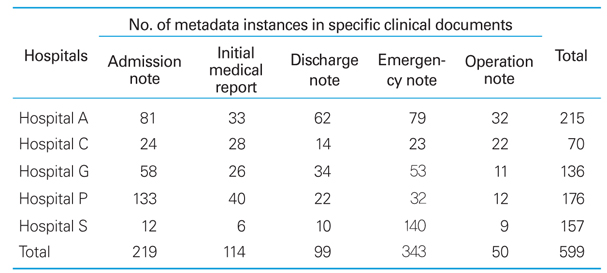
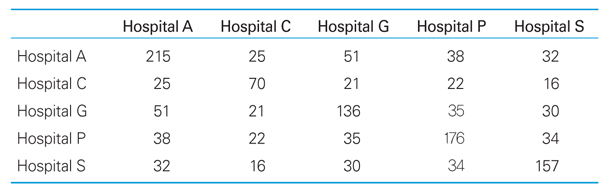
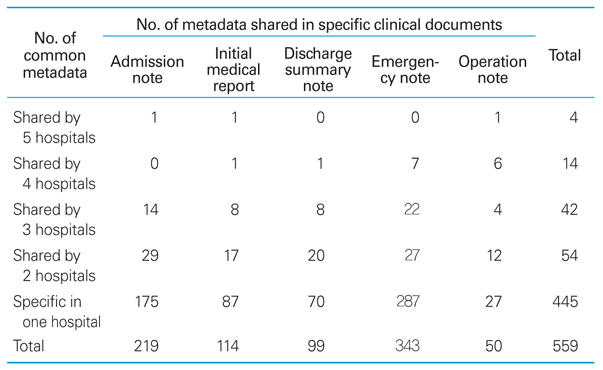
Distribution of metadata served by BMESH server
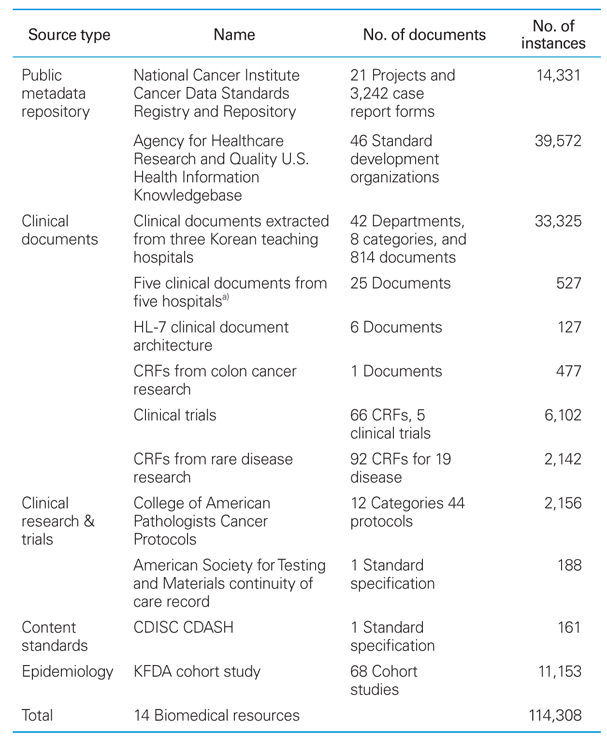
CRF, case report form; CDISC, Clinical Data Interchange Standards Consortium; CDASH, Clinical Data Acquisition Standards Harmonization ; KFDA, Korea Food & Drug Administration. a)Five clinical document types (admission note, initial medical examination note, discharge note, emergency note, and operation note) from five major Korean hospitals (Seoul National University Hospital, Ajou University Medical Center, Pusan National University Hospital, Gachon University Gil Hospital, and Chonnam National University Hospital).
- TOOLS