 |
 |
- Search
| J Korean Med Assoc > Volume 64(1); 2021 > Article |
|
Abstract
Lung cancer is the leading cause of cancer-related deaths in developing to advanced countries worldwide. The incidence rate of lung cancer in Korea has been increasing steadily since 1997. Statistics show that in 2017 alone, the incidence and mortality rates for lung cancer were 52.7 and 34.8 per 100,000 people, respectively, accounting for the highest cause of cancer death in Korea. The process of accurately diagnosing lung cancer consists of several steps, starting with the discovery of pulmonary nodule(s) via a cancer screening test or various other methods followed by the collection of cells or tissues and the identification of target molecules. Thereafter, staging and the development of a therapeutic plan lead to improved clinical outcomes. After the completion of a pilot study, a nationwide lung cancer screening program was introduced in Korea; since 2019, this program has targeted population at high risk for lung cancer: men and women aged 54 to 74 years who had a smoking history of 30 pack-years or more. The frequency of detection of pulmonary nodules is increasing in proportion to the public interest in health and economic growth. In this review, we present diagnostic techniques and biomarkers that are widely used in the medical field in the hope that such information would benefit clinical practice.
폐암은 개발도상국으로부터 선진국에 이르기까지 전 세계적으로 암 관련 사망의 주요 원인으로, 국내 폐암 발생률은 1997년부터 지속적으로 증가하는 추세를 보이고 있다[1]. 국내 통계에 의하면 2017년에는 10만 명당 52.7명의 발생률, 사망률은 10만 명당 34.8명으로 고형암 중에서 가장 높은 암 사망의 원인이다[1-3]. 이러한 대표적 다빈도 고위험 질환인 폐암에 대하여 국가폐암검진사업이 시범사업을 거쳐 2019년도부터 확대 시행되면서 폐 종괴의 발견과 함께 이의 정확한 진단에 대한 관심과 요구가 폭발적으로 늘어나고 있다[4]. 폐암이 진단되기까지는 영상을 이용한 폐 종괴의 발견, 발견된 종괴로부터 세포 또는 조직을 얻는 과정, 얻어진 검체로부터 암세포의 형태학적 확인과 바이오마커(biomarker)의 발견을 통한 치료 표적물을 선정하기까지 일련의 과정으로 구성되어 있다(Figure 1). 이번 고찰에서는 폐암을 진단하는 과정을 현재 의료현장에서 사용되고 있는 술기와 바이오마커 위주로 정리 기술하고자 하였다.
인류의 수명 연장, 흡연의 증가와 진단방법의 발전 등으로 인해 폐암의 발견이 점차 늘어나게 되고 따라서 폐암에 대한 선별검사들도 점차 많아지게 되었다. 1970년대 이후부터 시행되었던 객담 세포진 검사와 흉부 X선 촬영을 이용한 폐암의 선별검사는 낮은 민감도 등으로 인하여 대부분의 연구에서 대조군과 비교 시 피험군에서 폐암으로 인한 사망을 감소시키는데 실패하였다[5]. 후속으로 진행된 저선량 흉부 컴퓨터단층촬영(computed tomography, CT)를 이용하여 오랜 흡연력을 가진 폐암의 고위험군을 대상으로 선별검사를 실시하면 완치 가능한 상태의 조기 폐암 발견율을 높일 수 있고 이는 폐암으로 인한 사망률을 낮추는 것으로 이어질 수 있음이 확인됨으로써 전 세계적으로 폐암의 고위험군의 발견과 저선량 흉부 CT의 적용에 많은 관심을 가지게 되었다[6]. 이후 국내에서도 국가폐암검진 시범사업을 거쳐 2019년 국가암검진 프로그램에 폐암검진 프로그램을 도입하게 되었으며, 폐암 발생의 고위험군, 즉 만 54세 이상 만 74세 이하의 남녀 중 30갑년 이상의 흡연력을 가진 흡연자를 대상으로 2년 간격의 저선량 흉부 CT를 시행하고 있다. 저선량 흉부 CT는 16열 이상의 CT로 표준체중 수검자에서 방사선 선량을 CTDIvol 3 mGy 이하로 촬영해야 하며, 특정 교육을 이수한 영상의학과 전문의에 의해 촬영을 관리 감독하고 Lung Cancer Screening Reporting and Data System (Lung-RADS)에 근거한 판독을 시행하고 있다[7]. 발견된 결절은 성상 및 크기, CT 시행시기에 따라 범주 1-4의 결과로 분류하고, 이에 따라 침습적인 검사 혹은 CT 추적검사 등의 추가조치 및 금연교육을 시행한다. 검진의 결과는 이상소견 없음(범주 1), 양성 결절(범주 2), 경계성 결절(범주 3), 폐암 의심(범주 4A) 또는 폐암 매우 의심(범주 4B 또는 4X)의 범주로 나누어 보고된다. 2017년부터 2년간 시행된 시범사업 기간 동안 약 13,000명이 저선량 흉부 CT를 시행하였고, Lung-RADS에 근거한 판독 상 16%의 양성률을 보였으며, 발견된 폐암 중 조기 폐암은 60%에 달하였다. 이를 근거로, 저선량 흉부 CT를 이용한 폐암검진을 통한 조기 발견으로 폐암으로 인한 사망률을 감소시킬 것으로 기대되고 있다.
현재까지는 충분한 민감도, 특이도를 충족하는 폐암의 혈액 종양표지자가 없어, 폐암의 선별검사로서 혈액 종양표지자를 이용한 검사는 권고되지 않고 있으나, 혈액을 이용하는 검사들은 상대적으로 간단하고, 신속하며, 비침습적으로 실제 검진과 임상현장에서는 많이 시행되고 있다. 폐암에서 사용되고 있는 대표적인 선별을 위한 혈액 종양표지자는 다음과 같다.
대장암에서 흔히 사용되는 종양표지자이나, 유방암, 위암, 췌장암, 간암, 폐암에서도 증가할 수 있다. 폐암에서는 일반적으로 양성 예측율이 낮아 선별검사로 권고되지 않으나, 다양한 연구에서 혈청 carcinoembryonic antigen (CEA) 수치는 폐암의 예후와 재발 위험, 사망과 관련성을 높음을 보였다. 그러나 CEA는 암 이외에도 흡연, 신부전, 췌장염, 만성폐쇄성폐질환, 염증성장질환 등에서도 상승할 수 있으므로 해석에 주의를 요한다(참고치: 비흡연자인 경우 0-3 ng/mL, 흡연자인 경우 0-5 ng/mL) [8].
사이토 케라틴 19의 단편을 검출하는 것으로 비소세포 폐암, 특히 편평상피세포암에서 민감한 혈청 바이오마커로 알려져 있다[9]. Cytokeratin 19 fragment 21‑1 (CYFRA 21-1)은 폐암 이외에도 간, 위, 대장, 유방, 자궁경부의 암에서도 수치가 상승할 수 있다[8]. 폐암 진단에서 3.6 ng/mL의 cut-off 값을 사용하여, 52%의 민감도와 87%의 특이도를 가진다고 알려져 있으며[10], CYFRA 21-1 수치가 높은 경우 암의 진행 상태가 높고, 림프절 전이와 관련이 높은 것으로 알려져 있다. 종양 이외에도 신기능장애 및 만성폐쇄성폐질환, 간경변에서도 수치가 상승할 수 있어 해석에 주의를 요한다[11].
단백질 분해 효소 억제제의 일종으로, CYFRA 21-1보다는 비소세포암에서 덜 민감하나, 편평상피세포암에 대하여는 우수한 특이성을 가지고 있다[9]. CEA 종양표지자는 흡연에 영향을 받는 반면 squamous cell carcinoma‑related antigen (SCC-Ag)은 영향을 받지 않는 것으로 알려져 있으며, 과거 연구에서 CEA가 SCC-Ag보다 민감도가 높은 반면, SCC-Ag가 특이도가 높은 것으로 알려져 있다[12,13]. 폐암 이외에도, 자궁경부, 식도, 및 두경부 종양과 같은 다른 편평종양 및 피부질환, 신부전과 같은 양성질환에서도 상승할 수 있다.
신경내분비 기원의 종양 진단을 위한 바이오마커로 폐암 중 소세포암과 관련이 있다. 과거 연구에서 소세포 폐암 중 39-87%에서 neuron‑specific enolase (NSE)의 증가를 보고하였으며[14], NSE의 수치는 질병의 진행과 예후와 관련이 있다.
뇌, 폐, 결장 및 신경 내분비 세포에서 주로 발견되는 물질로, 소세포폐암에서 가장 민감하고 특이적인 종양표지자이다. 일반적으로 pro‑gastrin‑releasing peptide (proGRP)의 민감도가 NSE보다 높은 것으로 알려져 있다[15,16]. 84 ng/L의 cut-off로 폐암 이외에도 갑상선수질암 및 신장질환에서도 상승할 수 있다[9].
임상적으로 유용한 종양표지자의 양성률과 예측도는 폐암의 조직학적 특징에 따라 다르다. 현재 폐암의 종양표지자 측정에 대한 확립된 지침은 없으나, CEA, CYFRA 21-1, SCC-Ag, proGRP, NSE를 폐암의 예측에 포함할 수 있다. CEA, CYFRA 21-1, SCC-Ag는 비소세포폐암에 더 유용할 수 있으며, NSE과 proGRP의 조합은 소세포폐암의 진단에 유용할 수 있다. 다만 이러한 종양표지자의 사용은 비용적인 측면을 고려해야 한다.
폐암의 확인을 위한 조직학적 진단에 사용되는 검체로서는 객담, 기관지세척액, 기관지폐포세척액 등의 탈락세포를 이용하는 방법과 기관지 내시경검사, 세침흡인검사, 수술적 절제 등의 폐장 내 병변으로부터 직접적인 세포 획득을 통한 진단법이 있다. 조직검사는 폐암의 진단과 병기설정에 있어 필수적인데, 이를 통하여 병기 및 조직학적 성상, 유전자 및 분자생물학적 성상에 맞는 항암치료를 할 수 있다. 이러한 폐암의 조직학적 진단은 다시 비수술적인 조직검사와 수술적인 조직검사로 나뉘는데, 기관지 내시경검사와 세침흡인검사는 비수술적인 방법이며, 흉강경을 이용한 폐절제술은 수술적 방법에 해당한다.
기관지 내시경은 1897년 독일의 Gustav Killian이 처음 경직성 기관지 내시경을 발명하였고, 이후 일본의 Ikeda가 굴곡성 기관지 내시경을 발명함으로써 현재와 같이 널리 보급되게 되었다. 기관지 내시경에는 굴곡성 기관지 내시경검사, 투시유도 기관지 내시경검사, 기관지 초음파내시경, 내비게이션을 이용한 기관지 내시경검사 및 현재 개발 중인 로봇 기관지 내시경검사가 있다(Table 1). 원발성 병변의 위치가 중심형 폐암일 경우 굴곡성 기관지 내시경이 많이 사용되며, 말초 병변일 경우에는 투시유도 기관지 내시경, 방사형 기관지 초음파내시경, 내비게이션 기관지 내시경을 사용할 수 있다. 기관지 내시경 초음파 유도 하 세침흡인검사는 사용되는 탐색자의 종류에 따라 다시 방사형 탐색자(radial probe) 기관지 초음파내시경과 선형 탐색자(linear probe) 기관지 초음파내시경으로 구분되는데, 방사형 탐색자는 말초 병변의 조직검사에, 선형 탐색자는 중심 기관지 주변에 위치하는 림프선 조직검사에 사용된다. 이러한 기관지 내시경 초음파 유도하 세침흡인검사는 폐병변의 악성여부 진단에 있어서, 기존의 고식적인 경기관지 세침흡인검사 및 조영 흉부 CT, 양전자 방출 CT와 비교 시에 높은 민감도(92.3%)와 특이도(100%), 정확도(98%)를 가지는 것으로 보고되고 있다[17,18]. 최근 국내 Lee 등[19]이 보고한 바에 의하면 투시유도 기관지 내시경만 행하였을 경우 diagnostic yield가 59.4%였으나 방사형 기관지초음파를 같이 결합하여 사용했을 경우 diagnostic yield가 72.9%까지 향상됨을 보고하였으며, 57개의 연구들을 메타분석한 결과 방사형 기관지 초음파내시경의 diagnostic yield는 70.6% (49.4-92.3%)였다[19,20]. 폐암의 임파선 전이가 의심될 경우에는 선형 기관지 초음파내시경을 통해 전이 여부를 확인할 수 있다[21]. 로봇 기관지내시경에는 Monarch Platform (Auris Health, Redwood City, CA, USA)과 Ion Endoluminal System (Intuitive, Sunnyvale, CA, USA)이 있으며, 방사형 기관지초음파와 결합하여 diagnostic yield를 높일 수 있을 것으로 기대된다[22].
폐암의 진단에 있어서, 세침흡인검사 시 다양한 영상의학적 기법들을 활용하여 병변에 접근 가능한데, 주로 CT를 이용한 경피적 생검법이 많이 이용되고 있다. CT 유도하 세침흡인검사는 폐조직을 얻는데 가장 많이 사용되는 방법으로, 진단의 정확도는 병변의 크기와 연관이 있지만, 일반적으로 77%에서 94% 정도로 알려져 있다[21,23]. 몇몇 연구에서는 1 cm 이하의 병변에서도 그 진단적 유용성이 보고되었지만, 일반적으로 2 cm 이상의 말초 폐결절에서 높은 진단 정확도를 보인다[21].
폐의 세침흡인검사는 비교적 안전한 시술이지만, 종종 합병증이 뒤따르게 된다. Heerink 등[24]에 의한 최근의 메타분석연구에 따르면 경증의 합병증(특별한 처치가 불필요한 경증의 기흉, 경증의 폐출혈, 일시적인 객혈)은 19.6%에서, 중증의 합병증(중재적 처치가 필요한 중증의 기흉, 혈흉, 공기색전증, 파종성 전이, 사망)은 4.4%에서 발생하는 것으로 보고되었다. 이 중 가장 흔한 합병증은 기흉으로 전체 환자의 18.8%에서 발생하였고, 뒤를 이어 폐출혈(6.4%), 객혈(1.7%) 순으로 보고되었다. 또한 이 연구에서 이러한 합병증은 병변이 작을수록, 세침이 통과하는 폐실질이 길수록 더 많이 발생하는 것으로 보고하였다.
흉강경 폐절제술은 4-8 cm 크기의 절개창과, 두 개의 0.5 cm 크기의 포트, 그리고 늑간을 벌리지 않는 방법의 흉강수술을 통칭한다. 1994년 McKenna [25]가 흉강경을 이용한 폐엽절제술과 종격동 림프절절제술을 보고한 이후 흉강경을 이용한 폐절제술은 폐암의 진단 및 조기 폐암의 수술적인 치료 모두에 효과적인 방법으로 알려져 있는데 특히 종격동하부림프절(inferior mediastinal lymph node), 대동맥폐동맥창(aortopulmonary window lymph node), 대동맥주위림프절(para-aortic lymph node)의 조직검사에 선호되는 방법으로 동시에 위성 폐결절 및 흉막 또한 평가 가능한 장점이 있다[26].
일반적으로 폐병변의 생검을 위한 수술적 제거는 크게 두 가지 사항에서 고려되는데, 첫 번째는 수술 전 조직검사에서 양성 소견을 보이지만, 임상적으로 악성이 강하게 의심될 경우, 두 번째는 결절 자체가 수술 전 조직검사를 시도하지 않고 바로 절제를 선택할 수 있을 정도로 충분히 높은 위험으로 간주되는 경우이다. 악성이 의심될 경우 시행 의료기관별로 차이는 있지만, 일반적으로 권장되고 있는 적응증은 1기 비소세포암에서 폐엽절제술이 필요한 경우, 종양이 5-6 cm 이하인 경우, 기관지성 폐암이 아닌 경우, 일측폐 환기를 견딜 수 있는 경우로 제한되어 있다. 하지만 최근에는 그 적응증이 확대되어가고 있고, 특히 5 cm 이상의 비소세포성폐암에서도 효과적이라는 연구가 보고되고 있다[27]. 수술 후 경과에 있어, 흉강경 폐절제술을 개흉술과 비교했을 때, 입원 기간, 수술 후 폐기능, 운동기능에 있어 우수한 결과를 보이는 것으로 보고되고 있다. 특히 수술후의 통증의 측면에서도, 경막외 마취 거치기간이나 마약성 진통제 및 경구진통제의 사용량이 흉강경수술에서 유의하게 적은 것으로 보고되었다. 수술합병증으로 가장 흔한 합병증은 지속적인 폐공기 누출이며, 이는 폐의 장측 흉막에 손상이 있어 공기가 폐에서 흉강으로 누출되는 경우인데, 대부분 5일 이내에 호전되나, 7일 이상 지속 시 흉막유착술이 필요한 경우도 있다. 다른 합병증으로는 수술 후 발생하는 폐렴 등의 감염성 질환이 있다.
폐암의 진단은 크게 비소세포폐암과 소세포폐암을 포함하는 신경내분비종양으로 나뉜다. 먼저, 비소세포폐암은 크게 선암과 편평상피암으로 나눌 수 있는데, 이 둘을 구분하는 염색은 TTF-1, napsin A와 p40이 있다. TTF-1 및 napsin A는 선암의 표지자로써 이 두 마커 중 하나가 발현되면 선암으로 진단한다. 편평상피암에는 이 두 마커가 발현하지 않는다. p40의 경우는 편평상피암의 마커로써, 편평상피암으로 진단하려면 종양 전체에 p40이 100% 강하게 발현되어야 한다[28]. 간혹 분화가 나쁜 선암의 경우 p40을 발현할 수 있지만, 이러한 경우 국소적으로나마 TTF-1이나 napsin A를 발현할 수 있으며, 점액에 대한 mucicarmine 등의 특수염색에서 점액에 세포 두 개 이상에서 확인된다면 선암으로 진단할 수 있다[29]. 또한, p40이 종양세포 전체에 강하게 발현하지 않는다면, TTF-1과 napsin A가 음성이더라도 분화가 나쁜 선암으로 보는 것이 적절하다고 생각된다. 소세포폐암이 아니면서 모든 마커가 음성인 분화가 나쁜 비소세포폐암의 경우 대세포 폐암으로 진단하며 이는 반드시 수술적으로 절제된 조직에서만 가능한 진단이다.
신경내분비종양에는 전형적 카르시노이드(carcinoid), 비정형 카르시노이드, 대세포 신경내분비 암종, 소세포폐암이 포함된다[30]. 이들은 모두 신경내분비성 관련 염색인 synaptophysin, chromogranin A, CD56 등에서 양성 소견을 보이는 공통점을 가지며, 대세포 신경내분비 암종과 소세포폐암의 경우 선암과 같이 TTF-1을 발현할 수 있다. 이들의 감별은 조직학적 소견을 기반으로 우선적으로 이루어지는데, 간혹 대세포 신경내분비 암종과 소세포폐암의 감별이 어려운 경우 증식능을 알아보는 Ki67 염색을 한다면 소세포폐암에서는 증식능이 거의 100%에 육박하는데 반해 대세포 신경내분비 암종에서는 그보다 낮은 40-80% 정도를 보이게 된다[31].
진행성 폐암 진단 후 폐암의 유전자 변이를 확인하는 것은 치료에 필수적이다. 2000년대 후반 Gefitinib을 시작으로 폐암에서 표적치료제가 개발됨에 따라 수술적 치료가 불가능한 진행성 폐암에서 적절한 치료의 표적물을 찾는 것은 이제 폐암의 치료에서 필수적인 요건으로 되었다. Table 2는 현재 National Comprehensive Cancer Network 가이드라인에서 검사해야 할 분자진단 마커와 이에 대한 국내 빈도 및 이에 대한 항암제를 나타낸 표이다[32]. 현재 우리나라 의료현실에서 주로 사용 가능한 폐암의 분자 진단마커에는 EGFR, ALK, ROS1, BRAF가 있으며, 이에 대한 드라이버 돌연변이가 발견될 경우 이에 대한 경구 표적항암제를 일차적으로 사용할 수 있다. 이러한 표적항암제는 주로 하루 한 번 내지 두 번 먹는 형태이며, 부작용은 과거 전통적으로 사용했던 백금계 항암약물보다 적고, 효과는 더 커서, 과거보다 환자의 삶의 질이 많이 향상되었다. 그 외의 폐암 환자에 대해서는 MET exon 14 skipping mutation과 RET을 조사하여 각각의 환자에 맞는 표적항암제를 선택하게 된다. 표적 항암제의 사용이 여의치 않을 경우에는 면역관문억제제를 사용할 수 있으며, 이 경우 면역관문억제제의 바이오마커인 tumor mutation burden, PD-1 (programmed cell death 1, PDCD1), PD-L1 (programmed cell death 1 ligand 1, CD274)을 조사하게 된다. 하지만 tumor mutation burden 측정은 비싸고, 종양내 이질성을 대표하지 못하는 한계가 있기 때문에 일반적으로 PD-1, PD-L1을 측정하며, 측정된 PD-1, PD-L1 발현정도가 높을수록 면역관문억제제에 대한 효과가 좋을 것으로 기대한다[33,34]. CheckMate study, KEYNOTE study, OAK study, IMpower study는 진행성 폐암환자에서 면역관문억제제가 기존 항암제보다 의미 있게 무진행 생존기간과 총 생존기간이 증가하는 것을 보여주었으며, PACIFIC trial은 3기 비소세포폐암 환자에서 동시항암방사선 치료 후 durvalumab을 사용하였을 경우 의미 있게 무진행 생존기간과 총 생존기간이 증가함을 보여주었다. 최근에는 수술이 가능한 폐암 환자에서 수술 전 면역관문억제제의 사용에 대한 효과를 보는 연구들이 진행되고 있으며, 이에 대해 선행연구들은 긍정적인 효과를 보여, 후속 연구들의 결과가 기대되고 있다[35,36].
세포유리 DNA (cell-free DNA)는 혈액을 포함한 다양한 체액 내에 돌아다니는 작은 DNA로 건강한 사람에서는 낮은 범위의 농도(1-10 ng/mL)를 유지하나, 급성 외상, 뇌경색, 이식, 감염, 암과 같은 다양한 생태 병리학적 현상에서 증가할 수 있다. 특히 암 환자에서는 세포유리 DNA 중, 종양으로부터 유래된 순환 종양 DNA (circulating tumor DNA)에서 원발 종양세포가 보유한 동일한 돌연변이를 발견할 수 있다[37,38]. 액상생검(liquid biopsy) 기술은 악성 고형암 환자의 말초혈액에서 종양세포로부터 방출되는 유전자를 검사하는 방법으로 암세포의 다양한 유전적 변화를 순환 종양 DNA, 순환 종양 세포, 엑소좀(exosome), microRNA 또는 tumor educated platelet을 이용하여 검출하는 기술이다[37,39,40]. 국내에서 시행하는 고형암 액상생검은 혈액 및 체액 검체에서 세포유리 DNA를 추출하여, 순환 종양 DNA를 분석하는 검사로 악성종양의 치료 방침 결정 및 예후 판정에 도움을 줄 수 있으며, 폐암에서도 사용되고 있다[41,42]. 폐암의 침습적 조직검사가 적지 않은 확률로 합병증을 유발하고[20,23,43], 컨디션이 좋지 않은 고령의 환자가 많아, 액상생검은 비침습적 방법으로 안전하고, 반복적으로 검사할 수 있는 장점이 있다. 현재 European Medicines Agency와 US Food and Drug Administration에서는 비소세포폐암 환자 중 epidermal growth factor receptor (EGFR) 돌연변이를 조직생검 체취가 어려울 경우, 순환 종양 DNA 분석 기반으로 인정하였으며[44], 국내에서도 EGFR 돌연변이를 혈액을 포함한 액상생검 기법으로 진단하여 EGFR에 대한 표적치료제를 사용할 수 있도록 요양급여를 인정하였다.
이번 고찰을 통해 현재 폐암의 진단을 위해 의료현장에서 쓰이고 있는 다양한 진단도구에 대하여 살펴보았다. 폐장 내 종괴의 위치뿐 아니라 환자의 연령과 동반질환을 포함한 전신적 상태, 복용하고 있는 약제 등에 따라 최선의 진단방법이 무엇인지 시술을 선택하기 전에 고찰이 필요하다. 기술적 진보에 의해 이러한 진단도구들은 계속 발전과 소멸을 지속하고 있으나 폐암의 치료성적은 조기 발견, 나날이 개발되고 있는 적절한 약제의 올바른 적용을 통해 획기적으로 향상되고 있다. 이 논문에서 기술한 진단도구들은 현재 의료현장에서 폐암의 진단과 치료 과정에 보편적으로 널리 사용되고 있는 것들로서 적재 적소에 환자의 진료에 큰 도움이 될 수 있는 방법을 신중히 선택하고 적용하는 것이 필요하다.
Acknowledgement
This work was supported by the National Research Foundation of Korea (grant no. NRF-2020R1A2B5B01001883).
Figure 1.
Initial diagnostic workflow for lung cancer at clinics. CT, computed tomography; FBS, fiberoptic bronchoscope; EBUS, endobronchial ultrasound; NAB, needle aspiration biopsy; VATS, video-assisted thoracoscopic surgery; PET, positron emission tomography; MRI, magnetic resonance imaging; IHC, immunohistochemical staining; NGS, next generation sequencing; PCR, polymerase chain reaction.
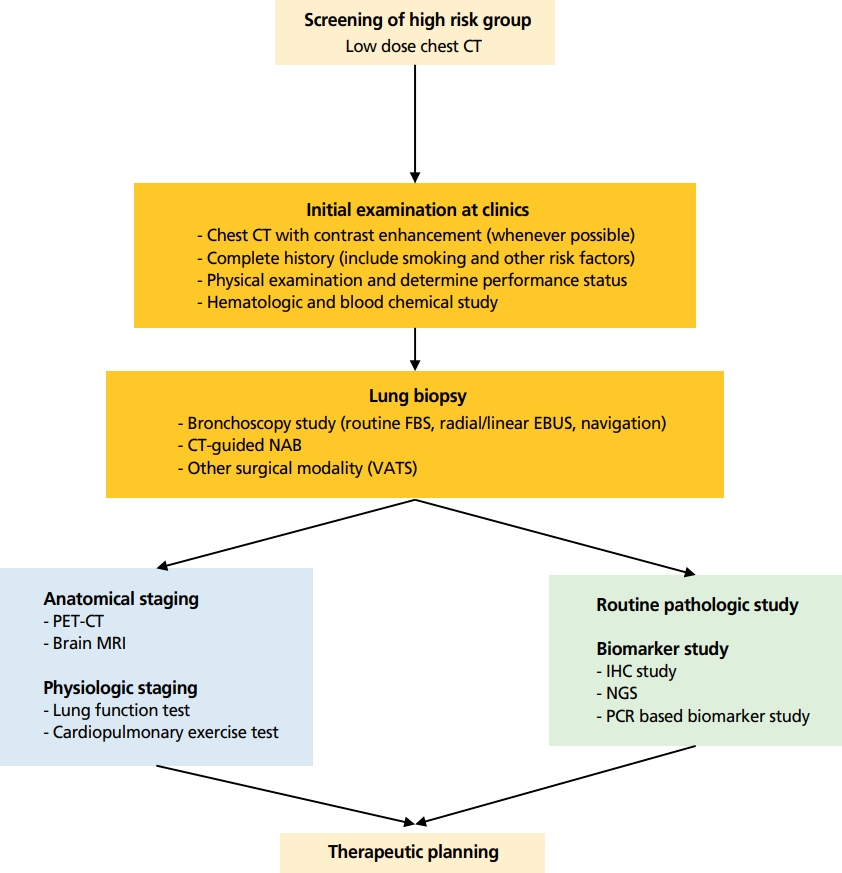
Table 1.
Types and roles of bronchoscopy
Table 2.
Molecular marker of lung cancer mainly used in clinical practice
References
1. Park CK, Kim SJ. Trends and updated statistics of lung cancer in Korea. Tuberc Respir Dis (Seoul) 2019;82:175-177.


2. Kim YC, Won YJ. The development of the Korean Lung Cancer Registry (KALC-R). Tuberc Respir Dis (Seoul) 2019;82:91-93.


3. Kim HC, Jung CY, Cho DG, Jeon JH, Lee JE, Ahn JS, Kim SJ, Kim Y, Kim YC, Kim JE, Lee B, Won YJ, Choi CM. Clinical characteristics and prognostic factors of lung cancer in Korea: a pilot study of data from the Korean Nationwide Lung Cancer Registry. Tuberc Respir Dis (Seoul) 2019;82:118-125.


5. Oken MM, Hocking WG, Kvale PA, Andriole GL, Buys SS, Church TR, Crawford ED, Fouad MN, Isaacs C, Reding DJ, Weissfeld JL, Yokochi LA, O'Brien B, Ragard LR, Rathmell JM, Riley TL, Wright P, Caparaso N, Hu P, Izmirlian G, Pinsky PF, Prorok PC, Kramer BS, Miller AB, Gohagan JK, Berg CD; PLCO Project Team. Screening by chest radiograph and lung cancer mortality: the Prostate, Lung, Colorectal, and Ovarian (PLCO) randomized trial. JAMA 2011;306:1865-1873.


6. National Lung Screening Trial Research Team, Aberle DR, Adams AM, Berg CD, Black WC, Clapp JD, Fagerstrom RM, Gareen IF, Gatsonis C, Marcus PM, Sicks JD. Reduced lung-cancer mortality with low-dose computed tomographic screening. N Engl J Med 2011;365:395-409.



7. Kim HY. National lung cancer screening in Korea: introduction and imaging quality control. J Korean Soc Radiol 2019;80:826-836.

8. Nakamura H, Nishimura T. History, molecular features, and clinical importance of conventional serum biomarkers in lung cancer. Surg Today 2017;47:1037-1059.



9. Duffy MJ, O'Byrne K. Tissue and blood biomarkers in lung cancer: a review. Adv Clin Chem 2018;86:1-21.


10. Pujol JL, Grenier J, Daures JP, Daver A, Pujol H, Michel FB. Serum fragment of cytokeratin subunit 19 measured by CYFRA 21-1 immunoradiometric assay as a marker of lung cancer. Cancer Res 1993;53:61-66.

11. Xu RH, Liao CZ, Luo Y, Xu WL, Li K, Chen JX, Huang YF, Chen YC, Zhu L, Yuan WB. Optimal cut-off values for CYFRA 21-1 expression in NSCLC patients depend on the presence of benign pulmonary diseases. Clin Chim Acta 2015;440:188-192.


12. Body JJ, Sculier JP, Raymakers N, Paesmans M, Ravez P, Libert P, Richez M, Dabouis G, Lacroix H, Bureau G, Thiriaux K, Lecomte J, Brohee D, Mommen P, Fruhling J, Klastersky J. Evaluation of squamous cell carcinoma antigen as a new marker for lung cancer. Cancer 1990;65:1552-1556.


13. Castelli M, Salvati F, Cruciani A, Portalone L, Giannarelli D, Ferrini U. Comparative analysis of CEA and SCC serum markers with IAP in human lung cancer. Int J Biol Markers 1989;4:45-50.


14. Carney DN, Marangos PJ, Ihde DC, Bunn PA Jr, Cohen MH, Minna JD, Gazdar AF. Serum neuron-specific enolase: a marker for disease extent and response to therapy of small-cell lung cancer. Lancet 1982;1:583-585.


15. Okusaka T, Eguchi K, Kasai T, Kurata T, Yamamoto N, Ohe Y, Tamura T, Shinkai T, Saijo N. Serum levels of pro-gastrin-releasing peptide for follow-up of patients with small cell lung cancer. Clin Cancer Res 1997;3:123-127.

16. Takada M, Kusunoki Y, Masuda N, Matui K, Yana T, Ushijima S, Iida K, Tamura K, Komiya T, Kawase I, Kikui N, Morino H, Fukuoka M. Pro-gastrin-releasing peptide (31-98) as a tumour marker of small-cell lung cancer: comparative evaluation with neuron-specific enolase. Br J Cancer 1996;73:1227-1232.




17. Herth F, Becker HD, Ernst A. Conventional vs endobronchial ultrasound-guided transbronchial needle aspiration: a randomized trial. Chest 2004;125:322-325.


18. Yasufuku K, Nakajima T, Motoori K, Sekine Y, Shibuya K, Hiroshima K, Fujisawa T. Comparison of endobronchial ultrasound, positron emission tomography, and CT for lymph node staging of lung cancer. Chest 2006;130:710-718.


19. Lee SC, Kim EY, Chang J, Lee SH, Han CH. Diagnostic value of the combined use of radial probe endobronchial ultrasound and transbronchial biopsy in lung cancer. Thorac Cancer 2020;11:1533-1540.



20. Moon SM, Choe J, Jeong BH, Um SW, Kim H, Kwon OJ, Lee K. Diagnostic performance of radial probe endobronchial ultrasound without a guide-sheath and the feasibility of molecular analysis. Tuberc Respir Dis (Seoul) 2019;82:319-327.



21. Park HJ, Lee SH, Chang YS. Recent advances in diagnostic technologies in lung cancer. Korean J Intern Med 2020;35:257-268.



22. Agrawal A, Hogarth DK, Murgu S. Robotic bronchoscopy for pulmonary lesions: a review of existing technologies and clinical data. J Thorac Dis 2020;12:3279-3286.



23. Jeon MC, Kim JO, Jung SS, Park HS, Lee JE, Moon JY, Chung CU, Kang DH, Park DI. CT-guided percutaneous transthoracic needle biopsy using the additional laser guidance system by a pulmonologist with 2 years of experience in CT-guided percutaneous transthoracic needle biopsy. Tuberc Respir Dis (Seoul) 2018;81:330-338.



24. Heerink WJ, de Bock GH, de Jonge GJ, Groen HJ, Vliegenthart R, Oudkerk M. Complication rates of CT-guided transthoracic lung biopsy: meta-analysis. Eur Radiol 2017;27:138-148.


25. McKenna RJ Jr. Thoracoscopic lobectomy with mediastinal sampling in 80-year-old patients. Chest 1994;106:1902-1904.


26. Howington JA. The role of VATS for staging and diagnosis in patients with non-small cell lung cancer. Semin Thorac Cardiovasc Surg 2007;19:212-216.


27. Batihan G, Ceylan KC, Usluer O, Kaya SO. Video-assisted thoracoscopic surgery vs thoracotomy for non-small cell lung cancer greater than 5 cm: is VATS a feasible approach for large tumors? J Cardiothorac Surg 2020;15:261.




28. Travis WD, Brambilla E, Noguchi M, Nicholson AG, Geisinger KR, Yatabe Y, Beer DG, Powell CA, Riely GJ, Van Schil PE, Garg K, Austin JH, Asamura H, Rusch VW, Hirsch FR, Scagliotti G, Mitsudomi T, Huber RM, Ishikawa Y, Jett J, Sanchez-Cespedes M, Sculier JP, Takahashi T, Tsuboi M, Vansteenkiste J, Wistuba I, Yang PC, Aberle D, Brambilla C, Flieder D, Franklin W, Gazdar A, Gould M, Hasleton P, Henderson D, Johnson B, Johnson D, Kerr K, Kuriyama K, Lee JS, Miller VA, Petersen I, Roggli V, Rosell R, Saijo N, Thunnissen E, Tsao M, Yankelewitz D. International association for the study of lung cancer/american thoracic society/european respiratory society international multidisciplinary classification of lung adenocarcinoma. J Thorac Oncol 2011;6:244-285.



29. Thunnissen E, Boers E, Heideman DA, Grunberg K, Kuik DJ, Noorduin A, van Oosterhout M, Pronk D, Seldenrijk C, Sietsma H, Smit EF, van Suylen R, von der Thusen J, Vrugt B, Wiersma A, Witte BI, den Bakker M. Correlation of immunohistochemical staining p63 and TTF-1 with EGFR and K-ras mutational spectrum and diagnostic reproducibility in non small cell lung carcinoma. Virchows Arch 2012;461:629-638.


30. Righi L, Gatti G, Volante M, Papotti M. Lung neuroendocrine tumors: pathological characteristics. J Thorac Dis 2017;9(Suppl 15):S1442-S1447.

31. Travis WD, Brambilla E, Burke AP, Marx A, Nicholson AG. WHO classification of tumours of the lung, pleura, thymus and heart. 4th ed. Lyon: International Agency for Research on Cancer; 2015.
32. El-Telbany A, Ma PC. Cancer genes in lung cancer: racial disparities: are there any? Genes Cancer 2012;3:467-480.



33. Kim HC, Choi CM. Current status of immunotherapy for lung cancer and future perspectives. Tuberc Respir Dis (Seoul) 2020;83:14-19.


34. Kim T, Cha YJ, Chang YS. Correlation of PD-L1 expression tested by 22C3 and SP263 in non-small cell lung cancer and its prognostic effect on EGFR mutation-positive lung adenocarcinoma. Tuberc Respir Dis (Seoul) 2020;83:51-60.



35. Forde PM, Chaft JE, Smith KN, Anagnostou V, Cottrell TR, Hellmann MD, Zahurak M, Yang SC, Jones DR, Broderick S, Battafarano RJ, Velez MJ, Rekhtman N, Olah Z, Naidoo J, Marrone KA, Verde F, Guo H, Zhang J, Caushi JX, Chan HY, Sidhom JW, Scharpf RB, White J, Gabrielson E, Wang H, Rosner GL, Rusch V, Wolchok JD, Merghoub T, Taube JM, Velculescu VE, Topalian SL, Brahmer JR, Pardoll DM. Neoadjuvant PD-1 blockade in resectable lung cancer. N Engl J Med 2018;378:1976-1986.



36. Cottrell TR, Thompson ED, Forde PM, Stein JE, Duffield AS, Anagnostou V, Rekhtman N, Anders RA, Cuda JD, Illei PB, Gabrielson E, Askin FB, Niknafs N, Smith KN, Velez MJ, Sauter JL, Isbell JM, Jones DR, Battafarano RJ, Yang SC, Danilova L, Wolchok JD, Topalian SL, Velculescu VE, Pardoll DM, Brahmer JR, Hellmann MD, Chaft JE, Cimino-Mathews A, Taube JM. Pathologic features of response to neoadjuvant anti-PD-1 in resected non-small-cell lung carcinoma: a proposal for quantitative immune-related pathologic response criteria (irPRC). Ann Oncol 2018;29:1853-1860.



37. Wan JC, Massie C, Garcia-Corbacho J, Mouliere F, Brenton JD, Caldas C, Pacey S, Baird R, Rosenfeld N. Liquid biopsies come of age: towards implementation of circulating tumour DNA. Nat Rev Cancer 2017;17:223-238.


38. Kang BJ, Ra SW, Lee K, Lim S, Son SH, Ahn JJ, Kim BC. Circulating tumor cell number is associated with primary tumor volume in patients with lung adenocarcinoma. Tuberc Respir Dis (Seoul) 2020;83:61-70.


39. Moon DH, Kwon SO, Kim WJ, Hong Y. Identification of serial DNA methylation changes in the blood samples of patients with lung cancer. Tuberc Respir Dis (Seoul) 2019;82:126-132.


40. Zheng H, Wu X, Yin J, Wang S, Li Z, You C. Clinical applications of liquid biopsies for early lung cancer detection. Am J Cancer Res 2019;9:2567-2579.


41. Lee SH, Kim EY, Kim T, Chang YS. Compared to plasma, bronchial washing fluid shows higher diagnostic yields for detecting EGFR-TKI sensitizing mutations by ddPCR in lung cancer. Respir Res 2020;21:142.




42. Kim T, Kim EY, Lee SH, Kwon DS, Kim A, Chang YS. Presence of mEGFR ctDNA predicts a poor clinical outcome in lung adenocarcinoma. Thorac Cancer 2019;10:2267-2273.



Peer Reviewers’ Commentary
이 논문은 폐암의 선별검사, 조직 진단과 치료에 이르는 전 과정의 폐암 진단법들에 대해 임상의가 알아야 할 부분을 중심으로 간결하고 체계적으로 기술하고 있다. 폐암은 국내 고형암 중 남녀 모두에서 암 사망률 1위를 차지할 정도로 예후가 불량하여 조기 진단이 매우 중요한 질환이다. 또한, 최근 개인 맞춤형 치료가 자리 잡으면서 적절한 치료법 선택을 위한 새로운 폐암 진단법들이 널리 이용되고 있다. 2019년부터 국가 폐암 검진사업이 시행되는 등 폐암 검사가 확대되고 다양화되는 현시점에서, 이 논문은 아주 시의적절하게 의료 현장에서 임상의가 알아야 할 폐암 진단법을 소개하고 있다. 이 논문은 임상의들이 현재 사용하고 있는 개별 폐암 진단법들을 이해하고, 이를 적용하는데 많은 도움이 될 것으로 판단된다.
[정리: 편집위원회]
-
METRICS

-
- 0 Crossref
- Scopus
- 6,843 View
- 351 Download
-
Related articles in
J Korean Med Assoc -
Transthoracic needle biopsy for diagnosis of lung cancer2023 March;66(3)
All-round counterattack to conquer lung cancer2023 March;66(3)
Evaluation and diagnostic approach for heart murmurs in children2020 July;63(7)
Causes and Diagnostic Strategies for Chronic Low Back Pain2007 June;50(6)






