|
 |
- Search
| J Korean Med Assoc > Volume 52(10); 2009 > Article |
Abstract
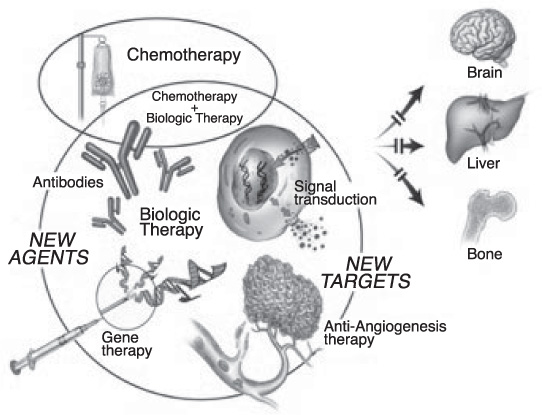
Breast cancer is the second common cancer and the leading cause of cancer deaths in women in Korea. Contrary to public perception, it is a heterogeneous disease with varying morphology, prognosis, and response to therapy. Understanding of the tumor biology results in marked advance in drug therapy proceeding to individualized molecular targeted therapy using predictive biomarkers (hormonal receptor: HR and human epidermal growth factor receptor -2: HER2). This review considers various drug therapeutic options based on biologic background of breast cancer divided into metastatic, neoadjuvant, and adjuvant systemic drug therapies. These are conventional cytotoxic chemotherapy, hormonal therapy, and molecular targeted therapies including trastuzumab, an anti-HER2 monoclonal antibody. In addition, bisphosphonate to improve outcomes of bone metastasis has seen an increased usage in adjuvant and metastatic setting. Microarray based genomic, transcription, and proteomic methods are transforming classification systems and identifying novel targets for the development of new therapeutics. It is important for us to appreciate and embrace the new developments as they will impact on daily clinical practice and require understanding of biomarkers as a tool for the determination of treatment options.
References
1. Buzdar AU. Endocrine therapy in the treatment of metastatic breast cancer. Semin Oncol 2001;28:291-304.
2. NCCN Practice Guidelines in Oncology-v.1. National Comprehensive Cancer Network Web site 2009;Accessed.
Breast/PDF/breast.pdf
3. Colozza M, de Azambuja E, Personeni N, Lebrun F, Piccart MJ, Cardoso F. Achievements in systemic therapies in the pregenomic era in metastatic breast cancer. Oncologist 2007;12:253-270.
4. Estevez LG, Tusquets I, Munoz M, Adrover E, Rovira PS, Segui MA, Rodriquez CA, Lescure AR, Ruiz M, Alvarez I, Mata JG. Advanced breast cancer: chemotherapy phase III trials that change a standard. Anticancer Drugs 2007;18:843-859.
5. Sledge GW, Neuberg D, Bernardo P, Ingle JN, Martino S, Rowinsky EK, Wood WC. Phase III trial of doxorubicin, paclitaxel, and the combination of doxorubicin and paclitaxel as front-line chemotherapy for metastatic breast cancer: An intergroup trial (E1193). J Clin Oncol 2003;21:588-592.
6. Nabholtz JM, Falkson C, Campos D, Szanto J, Martin M, Chan S, Pienkowski T, Zaluski J, Pinter T, Krzakowski M, Vorobiof D, Leonard R, Kennedy I, Azli N, Murawsky M, Riva A, Pouillart P. TAX 306 Study Group. Docetaxel and doxorubicin compared with doxorubicin and cyclophosphamide as first-line chemotherapy for metastatic breast cancer: Results of a randomized, multicenter, phase III trial. J Clin Oncol 2003;21:968-975.
7. Piccart MJ, Burzykowski T, Buyse M, Sledge G, Carmichael J, Luck HJ, Mackey JR, Nabholtz JM, Paridaens R, Biganzoli L, Jassem J, Bontenbal M, Bonneterre J, Chan S, Basaran GA, Therasse P. Taxanes alone or in combination with anthracyclines as first-line chemotherapy of patients with metastatic breast. J Clin Oncol 2008;26:1980-1986.
8. Gradishar WJ, Tjulandin S, Davidson N, Shaw H, Desai N, Bhar P, Hawkins M, O'Shaughnessy J. Phase III trial of nanoparticle albumin-bound paclitaxel compared with polyethylated castor oil-based paclitaxel in women with breast cancer. J Clin Oncol 2005;23:7794-7803.
9. Bonneterre J, Buzdar A, Nabholtz JM, Robertson JF, Thurlimann B, von Euler M, Sahmoud T, Webster A, Steinberg M. Arimidex Writing Committee. Investigators Committee Members. Anastrozole is superior to tamoxifen as first-line therapy in hormone receptor positive advanced breast cancer. Cancer 2001;92:2247-2258.
10. Nabholtz JM, Budzar A, Pollak M, Harwin W, Burton G, Mangalik A, Steinberg M, Webster A, von Euler M. Anastrozole is superior to tamoxifen as first-line therapy for advanced breast cancer in postmenopausal women: results of a North American multicenter trial. J Clin Oncol 2000;18:3758-3767.
11. Paridaens R, Beex LV, Nooij M, Cameron DA, Cufer T, Piccart MJ, Boqaerts J, Therasse P. the EORTC Breast Group. Phase III study comparing exemestane with tamoxifen as first-line hormonal treatment of metastatic breast cancer in postmenopausal women. J Clin Oncol 2008;26:4883-4890.
12. Mouridsen H, Gershanovich M, Sun Y, Perez-Carrion R, Boni C, Monnier A, Apffelstaedt J, Smith R, Sleeboom HP, Janicke F, Pluzanska A, Dank M, Becquart D, Bapsy PP, Salminen E, Snyder R, Lassus M, Verbeek JA, Staffler B, Chaudri-Ross HA, Dugan M. Superior efficacy of letrozole versus tamoxifen in postmenopausal women with advanced breast cancer: results of a phase III study of the international Letrozole Breast Cancer Group. J Clin Oncol 2001;19:2596-2606.
13. Mouridsen H, Gershanovich M, Sun Y, Perez-Carrion R, Boni C, Monnier A, Apffelstaedt J, Smith R, Sleeboom HP, Jaenicke F, Pluzanska A, Dank A, Becquart D, Bapsy PP, Salminen E, Snyder R, Chaudri-Ross H, Lang R, Wyld P, Bhatnagar A. Phase III study of letrozole versus tamoxifen as first-line therapy of advanced breast cancer in postmenopausal women: analysis of survival and update of efficacy from the international Letrozole Breast Cancer Group. J Clin Oncol 2003;21:2101-2109.
14. Rugo HS. The breast cancer continuum in hormone-receptor-positive breast cancer in postmenopausal women: evolving management options focusing on aromatase inhibitors. Ann Oncol 2008;19:16-27.
15. Howell A, Pippen J, Elledge RM, Mauriac L, Vergote I, Jones SE, Come SE, Osborne CK, Robertson JF. Fulvestrant versus anastrozole for the treatment of advanced breast carcinoma: a prospectively planned combined survival analysis of two multicenter trials. Cancer 2005;104:236-239.
16. Howell A, Robertson JF, Abram P, Lichinitser MR, Elledge R, Bajetta E, Watanabe T, Morris C, Webster A, Dimery I, Osborne CK. Comparison of fulvestrant versus tamoxifen for the treatment of advanced breast cancer in postmenopausalwomen previously untreated with endocrine therapy: a multinational, double-blind, randomized trial. J Clin Oncol 2004;22:1605-1613.
17. Klijn JG, Blamey RW, Boccardo F, Tominaga T, Duchateau L, Sylvester R. Combined tamoxifen and luteinizing hormone-releasing hormone (LHRH) agonist versus LHRH agonist alone in premenopausal advanced breast cancer: a meta-analysis of four randomized trials. J Clin Oncol 2001;19:343-353.
18. Slamon DJ, Leyland-Jones B, Shak S, Fuchs H, Paton V, Bajamonde A, Fleming T, Eiermann W, Wolter J, Pegram M, Baselga J, Norton L. Use of chemotherapy plus a monoclonal antibody against HER2 for metastatic breast cancer that overexpress HER2. N Engl J Med 2001;344:783-792.
19. Von Minckwitz G, du Bois A, Schmidt M, Maass N, Cufer T, de Jongh FE, Maartense E, Zielinski C, Kaufmann M, Bauer W, Baumann KH, Clemens MR, Duerr R, Uleer C, Andersson M, Stein RC, Nekljudova V, Liobl S. Trastuzumab beyond progression in human epidermal growth factor receptor 2-positive advanced breast cancer: a German breast group 26/breast international group 03-05 study. J Clin Oncol 2009;27:1999-2006.
20. Geyer CE, Forster J, Lindquist D, Chan S, Romieu CG, Pienkowski T, Jaqiello-Gruszfeld A, Crown J, Chan A, Kaufman B, Skarlos D, Campone M, Davidson N, Berqer M, Oliva C, Rubin SD, Stein S, Cameron D. Lapatinib plus capecitabine for HER2-positive advanced breast cancer. N Engl J Med 2006;355:2733-2743.
21. Moy B, Goss PE. Lapatinib-associated toxicity and practical management recommendations. Oncologist 2007;12:756-765.
22. Miller K, Wang M, Gralow J, Dickler M, Cobleigh M, Perez EA, Shenkier T, Cella D, Davidson NE. Paclitaxel plus bevacizumab versus paclitaxel alone for metastatic breast cancer. N Engl J Med 2007;357:2666-2676.
23. Goldhirsch A, Wood WC, Gelber RD, Coates AS, Thurlimann B, Senn HJ. Progress and promise: highlights of the international expert consensus on the primarytherapy of early breast cancer 2007. Ann Oncol 2007;18:1133-1144.
24. Goldhirsch A, Ingle JN, Gelber RD, Coates AS, Thurlimann B, Senn HJ. Thresholds for therapies: highlights of the St Gallen International Expert Consensus on the Primary Therapy of Early Breast Cancer 2009. Ann Oncol 2009;20:1319-1329.
25. Chia S, Norris B, Speers C, Cheang M, Gilks B, Gown AM, Huntsman D, Olivotto IA, Nielsen TO, Gelmon K. Human epidermal growth factor receptor 2 overexpression as a prognostic factor in a large tissue microarray series of node negative breast cancers. J Clin Oncol 2008;26:5697-5704.
26. Curigliano G, Viale G, Bagnardi V. Clinical relevance of HER-2 overexpression/amplification in patients with small tumor size (pT1a-b) and node-negative breast cancer. J Clin Oncol 2009;in press.
27. Smith IE. Targeting HER2 in the adjuvant setting: dealing with new standards and open questions. Breast 2009;18:Suppl 1. S17. (Abstr S41).
28. Buzdar AU, Singletary SE, Valero V, Booser DJ, Ibrahim NK, Rahman Z, Theriault RL, Walters R, Rivera E, Smith TL, Holmes FA, Hoy E, Frye DK, Manuel N, Kau SW, McNeese MD, Strom E, Thomas E, Hunt K, Ames F, Berry D, Hortobagyi GN. Evaluation of paclitaxel in adjuvant chemotherapy for patients with operable breast cancer: Preliminary data of a prospective randomized trial. Clin Cancer Res 2002;8:1073-1079.
29. Henderson IC, Berry DA, Demetri GD, Cirrincione CT, Goldstein LJ, Martino S, Ingle JN, Cooper MR, Hayes DF, Tkaczuk KH, Fleming G, Holland JF, Duggan DB, Carpenter JT, Frei E 3rd, Schilsky RL, Wood WC, Muss HB, Norton L. Improved outcomes from adding sequential paclitaxel but not from escalating doxorubicindose in an adjuvant chemotherapy regimen for patients with node-positive primary breast cancer. J Clin Oncol 2003;21:976-983.
30. Martin M, Pienkowski T, Mackey J, Pawlicki M, Guastalla JP, Weaver C, Tomiak E, Al-Tweiqeri T, Chap L, Juhos E, Guevin R, Howell A, Fornander T, Hainsworth J, Coleman R, Binholes J, Modiano M, Pinter T, Tang SC, Colwell B, Prady C, Provencher L, Walde D, Rodriguez-Lescure A, Hugh J, Loret C, Rupin M, Blitz S, Jacobs P, Murawsky M, Riva A, Voqul C. Breast Cancer International Research Group 001 Investigators. Adjuvant docetaxel for node-positive breast cancer. N Engl J Med 2005;352:2302-2313.
31. Goldstein LJ, O'Neill A, Sparano J. E2197. Phase III AT (doxorubicin/docetaxel) vAC (doxorubicin/cyclophosphamide) in the adjuvant treatment of node-positive and high risk node-negative breast cancer. J Clin Oncol 2005;23:16s. (suppl: abstract 512).
32. Martin M, Rodriguez-Lescure A, Ruiz A, Alba E, Calvo L, Ruiz-Borrego M, Munarriz B, Rodriguez CA, Crespo C, de Alava E, Lopez Garcia-Asenjo JA, Guitian MD, Almenar S, Gonzalez-Palacio S JF, Vera F, Palacios J, Ramos M, Gracia Marco JM, Lluch A, Alvarez I, Sequi MA, Mayordomo JI, Anton A, Baena JM, Palzaola A, Modolell A, Peleqri A, Mel JR, Aranda E, Adrover E, Alvarez JV, Garcia Puche JL, Sanchez-Rovira P, Gonzalez S, Lopez-Vega JM. Randomized phase III trial of fluorouracil, epirubicin, and cyclophosphamide alone or followed by paclitaxel for early breast cancer: GEICAM9906 Trial. J Natl Cancer Inst 2008;100:805-814.
33. Fountzilas G, Skarlos D, Dafni U, Gogas H, Briasoulis E, Pectasides D, Papadimitriou C, Markopoulos C, Polychronis A, Kalofonos HP, Siafaka V, Kosmidis P, Timotheadou E, Tsavdaridis D, Bafaloukos D, Papakostas P, Razis E, Makrantonakis P, Aravantinos G, Christodoulou C, Dimopoulos AM. Postoperative dose-dense sequential chemotherapy withepirubicin followed by CMF with or without paclitaxel in patients with high-risk operable breast cancer: A randomized phase III study conducted by the Hellenic Cooperative Oncology Group. Ann Oncol 2005;16:1762-1771.
34. Mamounas EP, Bryant J, Lembersky B, Fehrenbacher L, Sedlacek SM, Fisher B, Wickerham DL, Yothers G, Soran A, Wolmark N. Paclitaxel after doxorubicin plus cyclophosphamide as adjuvant chemotherapy for node-positive breast cancer: Results from NSABP B-28. J Clin Ocnol 2005;23:3686-3696.
35. Roche H, Fumoleau P, Spielmann M, Canon JL, Delozier T, Serin D, Symann M, Kerbrat P, Soulie P, Eichler F, Viens P, Monnier A, Vindevoqhel A, Campone M, Goudier MJ, Bonneterre J, Martin AL, Geneve J, Asselain B. Sequential adjuvant epirubicin-based and docetaxel chemotherapy for node-positive breast cancer patients. The FNCLCCPACS01 Trial. J Clin Oncol 2006;24:5664-5671.
36. The ATAC Trialists' Group. Results of the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial after completion of 5 years' adjuvant treatment of breast cancer. Lancet 2005;365:60-62.
37. The Arimidex, Tamoxifen Alone or in Combination Trialists' Group. Anastrozole alone or in combination with tamoxifen versus tamoxifen alone for adjuvant treatment of postmenopausal women with early breast cancer: First results of the ATAC randomized trial. Lancet 2002;359:2131-2139.
38. Goss PE, Ingle JN, Martino S, Robert NJ, Muss HB, Piccart MJ, Castiglione M, Tu D, Shepherd LE, Pritchard KI, Livingston RB, Davidson NE, Norton L, Perez EA, Abrams JS, Therasse P, Palmer MJ, Pater JL. A randomized trial of letrozole in postmenopausal women after five years of tamoxifen therapy for early stage breast cancer. N Engl J Med 2003;349:1793-1802.
39. Coombes RC, Hall E, Gibson LJ, Paridaens R, Jassem J, Delozier T, Jones SE, Alvarez I, Bertelli G, Ortmann O, Coates AS, Bajetta E, Dodwell D, Coleman RE, Fallowfield LJ, Mickewicz E, Andersen J, Lonning PE, Cocconi G, Stewart A, Stuart N, Snowdon CF, Carpentieri M, Massimini G, Bliss JM, van de Velde C. Intergroup Exemestane Study. A randomized trial of exemestane after two to three years of tamoxifen therapy in postmenopausal women with primary breast cancer. N Engl J Med 2004;350:1081-1092.
40. Jonat W, Grant M, Boccardo F. Switching from adjuvant tamoxifen to anastrozole in postmenopausal women with hormone-responsive early breast cancer: A meta-analysis of the ARNO 95 trial, ABCSG trial 8, and the ITA trial. Breast Cancer Res Treat 2005;94:11.
41. Jakesz R, Jonat W, Gnant M, Mittlboeck M, Greil R, Tausch C, Hilfrich J, Kwasny W, Menzel C, Samonigg H, Seifert M, Gademann G, Kauffmann M, Wolfgang J. ABCSG and the GABG. Switching of postmenopausal women with endocrine-responsive early breast cancer to anastrozole after 2 years' adjuvant tamoxifen: Combined results of ABSCG trial 8 and ARNO 95 trial. Lancet 2005;366:455-462.
42. Saphner T, Tormey DC, Gray R. Annual hazard rates of recurrence for breast cancer after primary therapy. J Clin Oncol 1996;14:2738-2746.
43. Smith I, Procter M, Gelber RD, Guillaume S, Feyereislova A, Dowsett M, Goldhirsch A, Untch M, Mariani G, Baselga J, Kaufmann M, Cameron D, Bell R, Bergh J, Coleman R, Wardley A, Harbeck N, Lopez RI, Mallmann P, Gelmon K, Wilchen N, Wist E, Sanchez Rovira P, Piccart MJ. HERA study team. 2-year follow-up of trastuzumab after adjuvant chemotherapy in HER2-positive breast cancer: a randomized controlled trial. Lancet 2007;369:29-36.
44. Romond EH, Perez EA, Bryant J, Suman VJ, Geyer CE Jr, Davidson NE, Tan-Chiu E, Martino S, Paik S, Maufman PA, Swain SM, Pisansky TM, Fehrenbacher L, Kutteh LA, Vogel VG, Visscher DW, Yothers G, Jenkins RB, Brown AM, Dakhil SR, Mamounas EP, Lingle WL, Klein PM, Ingle JN, Wolmark N. Trastuzumab plus adjuvant chemotherapy chemotherapy for operable HER2-positive breast cancer. N Engl J Med 2005;353:1673-1684.
45. Perez EA, Romond EH, Bryant J. Updated results of the combined analysis of NCCTG N9831 and NSABP B-31 adjuvant chemotherapy with/without trastuzumab in patients with HER2-positive breast cancer. J Clin Oncol 2007;24:18S. abstract 512.
46. Slamon D, Eiermann W, Robert N. BCIRG 006: 2nd interim analysis phase III randomized trial comparing doxorubicin and cyclophosphamide followed by docetaxel (AC→T) with doxorubicin and cyclophosphamide followed by docetaxel and trastuzumab (AC→TH) with docetaxel, carboplatin and trastuzumab (TCH) in HER2neu positive early breast cancer. Breast Cancer Res Treat 2006;100:abstract 52.
47. Joensuu H, Kellokumpu-Lehtinen P-L, Bono P, Alanko T, Kataja V, Asola R. Adjuvant docetaxel or vinorelbine with or without trastuzumab for breast cancer. N Engl J Med 2006;354:809-820.
48. Spielmann M, Roche H, Machiels JP. Trastuzumab following adjuvant chemotherapy in node-positive, HER2-positive breast cancer patients: 4-year follow-up results of the PACS-04 trial 2007;31th San Antonio Breast Cancer Symposium. San Antonio, Texas.
49. Wolmark N, Wang J, Mamounas E, Bryant J, Fisher B. Preoperative chemotherapy in patients with operable breast cancer: nine-year results from National Surgical Adjuvant Breast and Bowel Project B-18. Natl Cancer Inst Monogr 2001;30:96-102.
50. Kuerer HM, Newman LA, Smith TL, Ames FC, Hunt KK, Dhingra K, Theriault RL, Singh G, Binkely SM, Sneige N, Buchholz TA, Ross MI, McNeese MD, Buzdar AU, Hortobagyi GN, Singletary SE. Clinical course of breast cancer patients with complete pathologic primary tumor and axillary lymph node response to doxorubicin-based neoadjuvant chemotherapy. J Clin Oncol 1999;17:460-469.
51. Swain SM, Jeong JH, Geyer CE, Costantino JP, Pajon ER, Fehrenbacher L. abstract 75. NSABP B-30: definitive analysis of patient outcome from a randomized trial evaluating different schedules and combinationsof adjuvant therapy containing doxorubicin, docetaxel and cyclophophamide in women with operable, node-positive breast cancer 2008;32th San Antonio Breast Cancer Symposium. San Antonio, Texas.
52. Eiermann W, Paepke S, Appfelstaeldt J, Llombart-Cussac A, Eremin J, Vinholes J, Mauriac L, Ellis M, Chaudri-Ross HA, Dugan M, Borgs M. Letrozole Neo-Adjuvant Breast Cancer Study Group. Preoperative treatment of postmenopausal breast cancer patients with letrozole: A randomized double-blind multicenter study. Ann Oncol 2001;12:1527-1532.
53. Ellis MJ, Coop A, Singh B, Mauriac L, Llombert-Cussac A, Janicke F, Miller WR, Evans DB, Dugan M, Brady C, Quebe-Fehling E, Borgs M. Letrozole is more effective neoadjuvant endocrine therapy than tamoxifen for ErbB-1-and/or ErbB-2-positive, estrogen receptor-positive primary breast cancer: evidence from a phase III randomized trial. J Clin Oncol 2001;19:3808-3816.
54. Dowett M. on behalf of the ATAC Trialists' Group. Analysis of time to recurrence in the ATAC (Arimidex, Tamoxifen, Alone or in Combination) trial according to estrogen receptor and progesterone receptor status. Breast Cancer Res Treat 2003;83:abstract 143.
55. Smith I, Dowett M. on behalf of the IMPACT Trialists. Comparison of anastrozole vs tamoxifen vs tamoxifen alone and in combination as neoadjuvant treatment of estrogen receptor-positive (ER) operable breast cancer inpostmeno-pausal women the IMPACT trial. Breast Cancer Res Treat 2003;83:abstract 1.
56. Buzar AU, Ibrahim NK, Francis D, Booser D, Thomas ES, Theriault RL, Pusztai L, Arun BK, Giordano SH, Cristofanilli M, Frye DK, Smith TL, Hunt KK, Singletary SE, Sahin AA, Ewer MS, Buchholz TA, Berry D, Hortobagyi GN. Significantly higher pathological complete remission rate after neoadjuvant therapy with trastuzumab, paclitaxel, and epirubicin chemotherapy: results of a randomized trial in human epidermal group factor receptor 2-positive disease. J Clin Oncol 2005;23:3676-3685.
57. Buzar AU, Valero V, Ibrahim NK, Francis D, Broglio KR, Theriault RL, Pusztai L, Green MC, Singletary SE, Hunt KK, Sahin AA, Esteva F, Symmans WF, Ewer MS, Buchholz TA, Hortobagyi GN. Neoadjuvant therapy with paclitaxel followed by 5-flourouracil, epirubicin, and cyclophosphamide chemotherapy and concurrent trastuzumab in human epidermal growth factorreceptor 2-positive operable breast cancer: an update of the initial randomized study population and data of additional patients treated with the same regimen. Clin Cancer Res 2007;13:228-233.
58. Madarnas Y, Trudeau M, Franek JA, McCready D, Pritchard KI, Messersmith H. Adjuvant/neoadjuvant trastuzumab therapy in women with HER-2/neu-overexpressing breast cancer: A systemic review. Cancer Treat Rev 2008;34:539-557.
59. Kavanagh KL, Guo K, Dunford JE, Wu X, Knapp S, Ebetino FH, Rogers MJ, Russell RG, Oppermann U. The molecular mechanism of nitrogen-containing bisphophonates as anti-osteoporosis drugs. Proc Natl Acad Sci USA 2006;103:7829-7834.
60. Rogers MJ, Gordon S, Benford HL, Coxon FP, Luckman SP, Monkkonen J, Frith JC. Cellular and molecular mechanisms of action of bisphosphonates. Cancer 2000;88:2961-2978.
61. Corey E, Brown LG, Quinn JE, Poot M, Roudier MP, Higano CS, Vessella RL. Zoledronic acid exhibits inhibitory effects on osteoblastic and osteolytic metastases of prostatic cancer. Clin Cancer Res 2003;9:295-306.
62. Neville-Webbe HL, Rostami-Hodjegan A, Evans CA, Coleman RE, Holen I. Sequence and schedule-dependent enhancement of zoledronic acid induced apoptosis by doxoru-bicin in breast and prostate cancer cells. Int J Cancer 2005;113:364-371.
63. Ottewell PD, Jones M, Lefley DV, Coleman RE, Holen I. Combined effects of cytotoxic drugs and anti-resorptive agents in vivo. Breast Cancer Res Treat 2006;abstract 6102.
64. Van Valckenborgh E, De Raeve H, Devy L, Blacher S, Munaut C, Noël A, Van Marck E, Van Riet I, Van Camp B, Vanderkerken K. Murine 5T multiple myeloma cells induce angiogenesis in vitro and in vivo. Br J Cancer 2002;86:796-802.
65. Hillner BE, Ingle JN, Chlebowski RT, Gralow J, Yee GC, Janjan NA, Cauley JA, Blumenstein BA, Albain KS, Lipton A, Brown S. American Society of Clinical Oncology. American Society of Clinical Oncology 2003 update on the role of bisphosphonates and bone health issues in women with breast cancer. J Clin Oncol 2003;21:4042-4057.
66. Theriault RL, Biermann JS, Brown E, Brufsky A, Demers L, Grewal RK, Guise T, Jackson R, McEnery K, Podoloff D, Ravdin P, Shapiro CL, Smith M, Van Poznak CH. NCCN Task Force Report: Bone Health and Cancer Care. J Natl Compr Canc Netw 2006;4:1-20.
67. Hortobagyi GN, Theriault RL, Lipton A, Porter L, Blayney D, Sinoff C, Wheeler H, Simeone JF, Seaman JJ, Knight RD, Heffernan M, Mellars K, Reitsma DJ. Protocol 19 Aredia Breast Cancer Study Group. Long-term prevention of skeletal complications of metastatic breast cancer with pamidronate. J Clin Oncol 1998;16:2038-2044.
68. Theriault RL, Lipton A, Hortobagyi GN, Leff R, Gluck S, Stewart JF, Costello S, Kennedy I, Simeone J, Seaman JJ, Knight RD, Mellars K, Heffernan M, Reitsma DJ. Protocol 18 Aredia Breast Cancer Study Group. Pamidronate reduces skeletal morbidity in women with advanced breast cancer and lytic bone lesions: a randomized, placebo-controlled trial. J Clin Oncol 1999;17:846-854.
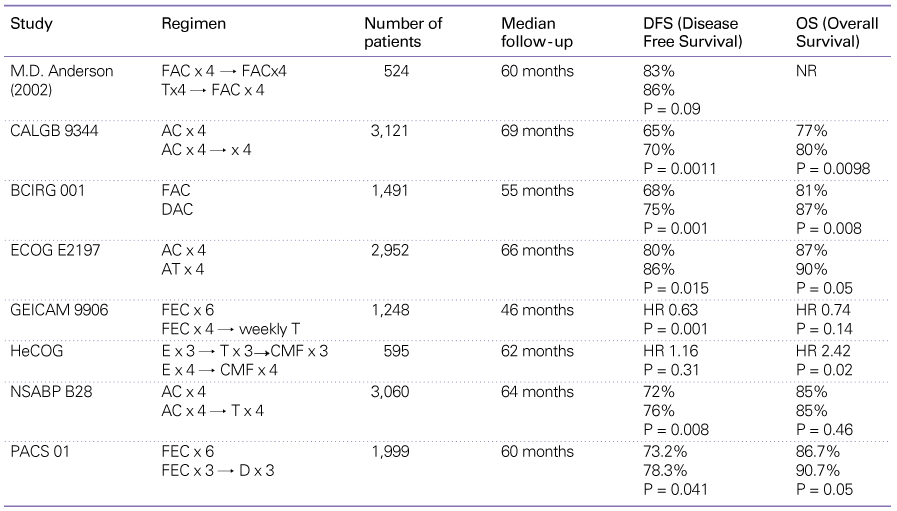
Table 3
Summary of trials comparing chemotherapy plus trastuzumab with chemotherapy alone in patients with metastatic breast cancer

Table 7
Summary of randomized, phase III trials evaluating adjuvant Trastuzumab in patients with HER2-positive early-stage breast cancer

T: trastuzumab, CTx: chemotherapy, AC: doxorubicin+cyclophosphamide, P: paclitaxel, DCT: docetaxel+carboplatin+ trastuzumab, V/D: vinorelbine or docetaxel, CEF: cyclophosphamide+epirubicin+fluorouracil, FECHR: hazard ratio, DFS: disease-free survival, DFSR: disease free survival rate, OS: overall survival, OSR: overall survival rate
- TOOLS