|
 |
- Search
| J Korean Med Assoc > Volume 46(1); 2003 > Article |
Abstract
Lung cancer is the leading cause of cancer deaths. Non-small cell lung cancer constitutes approximately 75% of lung cancers, and 40% present as advanced stage IIIa or IIIb, where primary surgery is ineffective. Radiation of the primary tumor and the regional lymphatics has been the traditional treatment for unresectable locally advanced disease, but few patients achieved a complete response. Because of the limited benefits provided by radiation therapy, the use of combined chemoradiotherapy in patients with locally advanced, unresectable NSCLC was explored. Combined chemoradiotherapy appears to have improved the outcome of patients with locally advanced unresectable stage III NSCLC, with a median survival of 13 to 14 months and 5-year survival rates as high as 15% to 20%, nearly three times that reported with radiation therapy alone and various agents have been used either sequentially or concomitantly in clinical trials of combined chemoradiotherapy for NSCLC. The interactions of chemotherapy and radiation therapy are complex. The texanes interact with radiation at many levels. Cell-cycle synchronization through mitotic arrest has been consistently shown to have a major role in radiation enhancement, but increased apoptosis and tumor reoxygenation may constitute additional mechanisms. Clearly, the interaction is multifactorial and the dominant mechanism may be affected by specific settings that include drug exposure and concentration, tumor type, and radiation dosimetry.
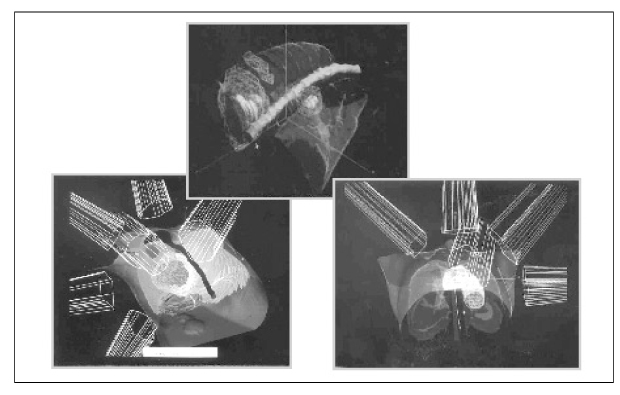
Recent studies have demonstrated that shorter high-dose radiotherapy schedules cause a statistically significant increase in local tumor control in NSCLC. Radiation dose escalation utilizing conventional fractionation techniques would likely cause prohibitive toxicity. Three-dimensional conformal radiation therapy (3-DCRT) had the potential to deliver high dose radiation (>70 Gy) with minimal underdosing and with a concomitant relative sparing of normal tissues. This technical demonstration of enhanced therapeutic ratio is the base for the evolving clinical utilization of 3-DCRT for NSCLC. Preliminary experience has resulted in promising survival following three-dimensional conformal radiation therapy alone for locally advanced NSCLC. More follow-up and experience will determine late toxicity, maximum dose, and efficacy of dose escalation with three-dimensional conformal radiation therapy. Strategies should be developed to integrate this modality into the combined treatment of locally advanced NSCLC.
Developments in biotechnology within the last decade have resulted in identification of important biological and biophysiological pathways in the lung carcinogenesis, and new agents are developed to target difficult levels of these important pathways.
Preclinical and clinical studies with these specific targeted therapies in lung cancer have been very promising. Targeted therapies in lung cancer and the potential of combining these agents with chemotherapy and radiotherapy are under investigation.
References
1. Bae JM, Won YJ, Jung KW, Suh KA, Ahn DH, Park JG. Annaual report of the central cancer registry in Korea-1999 : Based on registered data from 128 hospitals. Cancer Res Treat 2001;33(5):367-372.
3. Perez CA, Stanley K, Rubin P, et al. Preliminary report by the Radiation Therapy Oncology Group. A prospective randomized study of various irradiation doses and fractionation schedules in the treatment of inoperable non-oat cell carcinoma of the lung. Cancer 1980;45:2744-2753.
4. Perez CA, bauer M, Edelstein S, et al. Impact of tumor control on survival in carcinoma of the lung treated with irradiation. Int J Radiat Oncol Biol Phys 1986;12:539-547.
5. Perez CA, Pajak TF, Rubin P, et al. Long-term observations of the patterns of failure in patients with unresectable non-oat cell carcinoma of the lung treated with definitive radiotheapy. Cancer 1987;59:1874-1881.
6. Dillman RO, Seagren SL, Propert KJ, et al. A randomized trial of induction chemotherapy plus high-dose radiation versus radiation alone in stage III non-small-cell lung cancer. N Engl J Med 1990;323:940-945.
7. Arriagada R, Le Chevalier T, Quoix E, et al. ASTRO(American Society for Therapeutic Radiology and Oncology) pleanary: Effect of chemotherapy on locally advanced non-small cell lung carcinoma: a randomized study of 353 patients. Int J Radiat Oncol Biol Phys 1991;20:1183-1190.
8. Cox JD, Azarnia N, Byhardt RW, et al. A randomized phase I/II trial of hyperfractionated radiation therapy with total dose of 60.0Gy to 79.2Gy: Possible survival benefit with ≥ 69.6Gy in favorable patients with Radiation Therapy Oncology group stage III nonsmall cell carcinoma of the lung. Report of RTOG 83-11. J Clin Oncol 1990;8:1543-1555.
9. Byhardt RW, Martin L, Pajak TF, et al. The influence of field size and other treatment factors on pulmonary toxicity following hyperfractionated irradiation for inoperable nonsmall cell lung cancer(NSCLC)-Analysis of a Radiation Therapy Oncology Group(RTOG) protocol. Int J Radiat Oncol Biol Phys 1993;27:537-544.
10. Le Chevalier T, Arriagada R, Quoix E, et al. Radiotherapy alone versus combined chemotherapy and radiotheapy in unresectable nonsmall cell lung carcinoma. Lung Cancer 1994;10:S239-S252.
11. Dillman RO, Herndon J, Seagren SL, Eaton WL Jr, Green MR. Improved survival in stage III non-small-cell lung cancer: sevenyear follow-up of cancer and leukemia group B (CALGB) 8433 trial. J Natl Cancer Inst 1996;88:1210-1215.
12. Sause W, Kolesar P, Taylor S, et al. Final results of phase III trial in regionally advanced unresectable non-small cell lung cancer. Chest 2000;117:358-364.
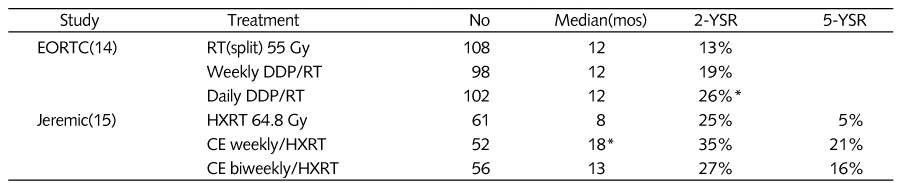
14. Schaake-Koning C, Van Den bogert W, Dalesio O, et al. Effects of concomitant cisplatin and radiotherapy in inoperable non-small cell lung cancer. N Engl J Med 1992;326:524-530.
15. Jeremic B, Shibamoto Y, Acimovic L, Djuric L. Randomized trial of hyperfractionated radiation therapy with or without concurrent chemotherapy for stage III non-small-cell lung cancer. J Clin Oncol 1995;13:452-458.
17. Furuse K, Fukuoka M, Kawahara M, et al. Phase III study of concurrent versus sequential thoracic radiotherapy in combination with Mitomycin, Vindesine, and Cisplatin in unresectable stage III non-small-cell lung cancer. J Clin Oncol 1999;17:2692-2699.
18. Komaki R, Seiferheld W, Curran W, et al. Sequential vs concurrent chemotherapy and radiation therapy for inoperable non-small cell lung cancer (NSCLCa): Analysis of failures in a phase III study (RTOG 9410). Int J Radiat Oncol Biol Phys 2000;48(3):suppl 1. 113.
19. Saunders M, Dische S, Barrett A, et al. Continuous hyperfractionated accelerated radiotheapy(CHART) versus conventional radiotherapy in non-small cell lung cancer: A randomized multicentre trial. Lancet 1997;350:161-165.
20. Tannehill S, Mehta M, Froseth C, et al. Phase II trial of hyperfractionated accelerated radiation therapy(HART) for unresectable non-small cell lung cancer(NSCLC): Preliminary results of ECOG 4593. Proc Am Soc Clin Oncol 1997;16:446a. (abstr).
21. Fletcher G. Clinical dose-response curves of human malignant epithelial tumours. Br J Radiol 1973;46:1-12.
22. Emami B, Purdy J, Harms W, et al. Three dimensional treatment planning for lung cancer. Int J Radiat Oncol Biol Phys 1991;21:217-227.
23. Graham MV, Matthews JW, Harms WB, et al. 3-D radiation treatment planning study for patients with carcinoma of the lung. Int J Radiat Oncol Biol Phys 1994;29:1105-1117.
24. Amstrong J, Raben A, Zeldfsky M, et al. Promising survival with three-dimensional conformal radiation therapy for non-small lung cancer. Radiother Oncol 1997;44:17-22.
25. Emami B, Scott C, Byhardt R, et al. The value of regional nodal radiotherapy(dose/volume) in the treatment of unresectable non-small cell lung cancer: an RTOG analysis. Proceedings of the 38th annual ASTRO meeting 1996;101.
26. Robertson J, Haken R, Hazuka M, et al. Dose escalation for non-small cell lung cancer using conformal radiation therapy. Int J Radiol Oncol Biol Phys 1997;37:1079-1085.
- TOOLS
-
METRICS

-
- 1 Crossref
- Scopus
- 1,128 View
- 4 Download
-
Related articles in
J Korean Med Assoc -
New targeted therapies for non-small cell lung cancer2023 March;66(3)
Robotic surgery for colorectal cancer2022 September;65(9)
Chemotherapy for Colorecal Cancer2002 July;45(7)
Role of Radiotherapy in High-Risk Colon Cancer2002 July;45(7)